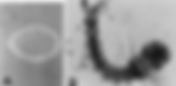197 results found with an empty search
- Agricultural Benefits of Lactiplantibacillus Plantarum in Biofertilizers
Lactiplantibacillus Plantarum , a lactic acid bacterium, is gaining ground in biofertilizers for its ability to improve soil health, unlock nutrients, and support crops under real-world farming challenges. pmc.ncbi.nlm.nih+1 Farmers use it in microbial blends to tackle issues like nutrient lockup, poor organic matter breakdown, and soil fatigue from years of synthetic fertilizers. This article focuses on its key agricultural benefits, with practical examples from field conditions. 1. Boosting Soil Health and Organic Matter Breakdown 1.1 Faster decomposition of residues and manures In biofertilizers, L. plantarum kick-starts fermentation of crop residues, manures, and green wastes—much like it does in silage or bokashi compost. gardenculturemagazine+1 Real farming example On a wheat field with heavy stubble residue, applying a LAB-containing biofertilizer (e.g., via soil drench) helps break down the leftover straw faster. This releases nitrogen and other nutrients tied up in the plant material, making them available for the next crop. It also reduces the risk of residue-borne diseases by creating a low-pH environment that favors beneficial microbes over pathogens. publishing.emanresearch+1 Benefits in tough conditions In compacted or clay-heavy soils, it improves organic matter turnover, leading to better crumb structure and water infiltration over time. Reduces odors and ammonia loss from manure applications, keeping more nitrogen in the field. seedandknowledge+1 Studies on LAB in sustainable agriculture confirm that L. plantarum speeds up composting by 20–50% and enhances humus formation, which supports long-term soil fertility. pmc.ncbi.nlm.nih+1 1.2 Building a more resilient soil microbiome L. plantarum doesn’t dominate the soil forever, but it helps reshape the microbial community toward better balance. Field impact After repeated applications in vegetable rotations, soils show increased populations of helpful groups like Bacillus and actinobacteria, while opportunistic pathogens decline. This creates “suppressive soils” that naturally resist issues like root rots. agritechinsights+1 In saline or over-fertilized fields, it helps restore diversity lost from chemical overuse, making the soil more forgiving during dry spells or wet seasons. 2. Improving Nutrient Availability 2.1 Solubilizing phosphorus and micronutrients L. plantarum produces organic acids (lactic, acetic) that lower pH locally around roots and residues, dissolving fixed phosphorus and micronutrients. sciencedirect+1 Practical crop example Tomato growers in calcareous (high-pH) soils apply LAB biofertilizers at transplanting. The acids from L. plantarum free up phosphorus bound to calcium, boosting early root growth and fruit set. Field trials show 10–25% more available P after LAB treatments, translating to healthier plants with less yellowing. agritechinsights+1 Wheat and cereals In P-deficient paddocks, seed treatments with L. plantarum increase root length by up to 2.4 times, helping seedlings tap into otherwise unavailable reserves.[ agris.fao ] 2.2 Enhancing nitrogen use from organic sources While it doesn’t fix nitrogen, L. plantarum accelerates the breakdown of organic N (from manures or cover crops) into plant-available forms. Livestock manure scenario Dairy farmers mix LAB blends into slurry before spreading. This minimizes N loss as gas and improves incorporation into soil, where it feeds crops more efficiently. Combined with reduced synthetic N rates, yields stay steady while costs drop. publishing.emanresearch+1 3. Supporting Crop Growth and Stress Tolerance 3.1 Better germination and root development L. plantarum forms protective biofilms on seeds and roots, promoting vigorous early growth. agris.fao+1 Seed treatment example Wheat farmers coat seeds with a biofertilizer containing L. plantarum before drilling. Germination rates improve by 6–40%, and seedlings emerge taller and stronger, even in cold, wet springs. Root systems expand faster, giving plants a head start against weeds and early droughts.[ agris.fao ] 3.2 Help with drought and heat stress Under stress, L. plantarum boosts plant antioxidants and maintains photosynthesis. Corn or soybean field In rainfed areas, foliar or soil-applied LAB helps crops hold more chlorophyll and activate enzymes like catalase. Plants stay greener longer, preserving yield during dry periods—critical for farmers facing unpredictable weather.[ jksus ] 3.3 Silage and forage quality for livestock systems In mixed crop-livestock farms, L. plantarum as a silage inoculant preserves more nutrients in grass or legume haylage. Outcome Soybean or amaranth silage treated with L. plantarum has higher lactic acid, lower pH, and better protein retention. Animals eat better feed, produce more milk or gain weight faster, and farmers save on supplements. frontiersin+1 4. Natural Pathogen Suppression in the Field 4.1 Reducing soil-borne diseases The acids and antimicrobial compounds from L. plantarum create zones around roots where pathogens struggle. Root rot example In potato or tomato rotations prone to Fusarium or Ralstonia, biofertilizer drenches with L. plantarum shift the soil community. Pathogen levels drop, and healthy root mass increases by 20–30%, leading to fewer skips and better stands. pmc.ncbi.nlm.nih+1 Integrated approach Works best alongside crop rotation, residue management, and other biologicals like Trichoderma—common in biofertilizer programs. 4.2 Post-harvest and storage support Sprays on fruits and vegetables extend shelf life by suppressing molds and bacteria, reducing losses for market growers. frontiersin+1 5. How Farmers Get Started with L. plantarum Biofertilizers 5.1 Choosing the right product Look for blends listing lactic acid bacteria (LAB) or L. plantarum specifically, at 10^8 CFU/ml or higher. Examples include EM-style activators from companies like IndoGulf BioAg. indogulfbioag+1 5.2 Application tips for real results Application When to Use Rate/Example Expected Benefit Seed treatment Pre-planting Dip or coat seeds +10–40% germination, stronger roots[ agris.fao ] Soil drench/drip Transplant or early tiller 1–5 L/ha diluted Nutrient unlock, disease suppression[ agritechinsights ] Compost/manure mix Before spreading 1:100 dilution Faster breakdown, less N loss[ pmc.ncbi.nlm.nih ] Foliar spray Vegetative stage 1–2 L/ha Stress tolerance, minor disease control[ jksus ] Key rules Apply with moisture for activation. Store cool and use within shelf life (check label). Pair with organics for best effect; reduce chemicals gradually. 5.3 Measuring success on your farm Track soil tests (available P, organic matter), crop vigor (root mass, chlorophyll), and yields. Improvements often show in 1–2 seasons as soil biology builds. 6. Why It Matters for Modern Farming In an era of rising input costs, climate variability, and regulations on chemicals, L. plantarum in biofertilizers offers practical, low-risk benefits: healthier soils, better nutrient efficiency, and more resilient crops. publishing.emanresearch+1 It shines in organic transitions, saline/chalky soils, and livestock-integrated systems. For deeper details on its functions and industry fit, check the pillar page “Lactiplantibacillus plantarum: Benefits, Functions, and Characteristics Across Industries.”
- What Are the Uses of Lactiplantibacillus Plantarum?
Photo credit: https://www.nature.com/articles/s41598-025-06103-9/figures/1 Lactiplantibacillus Plantarum (often still called Lactobacillus plantarum) is a friendly lactic acid bacterium best known from yogurt, pickles, and other fermented foods. Today, it is also an important “workhorse microbe” in agriculture, biofertilizers, soil health solutions, animal feed, and food processing. frontiersin+1 This article walks through its main practical uses with simple examples. For a fuller overview of how this microbe behaves in soil and across industries, see the pillar page “Lactiplantibacillus plantarum: Benefits, Functions, and Characteristics Across Industries.” 1. Helping Crops Grow: Uses in Agriculture and Biofertilizers 1.1 As a biofertilizer ingredient Many modern biofertilizers and microbial blends include L. plantarum alongside Bacillus, yeasts, and other beneficial microbes. indogulfbioag+1 How it’s used in the field Soil drench or through drip: Growers dilute a liquid microbial blend containing L. plantarum and apply it via irrigation. With organic fertilizers or compost teas: LAB-based products are mixed into organic inputs so they break down faster and release nutrients more steadily. gardenculturemagazine+1 What it does in simple terms Helps decompose organic matter, like crop residues and manures, making nutrients easier for roots to absorb. pmc.ncbi.nlm.nih+1 Produces natural acids that can “unlock” bound phosphorus and micronutrients in the soil. publishing.emanresearch+1 Competes with harmful microbes around the root, contributing to a healthier rhizosphere. 1.2 In multi-strain microbial blends (“EM-type” products) L. plantarum is a core member of many “Effective Microorganisms”‑style products. For example, microbial blends like Micro-Manna include lactic acid bacteria (including L. plantarum), photosynthetic bacteria, and yeasts at about 1 × 10^8 CFU/ml. indogulfbioag+1 Practical uses Soil revitalization: Applied to tired, compacted, or saline soils to rebuild biological activity and improve structure over time. Crop kits: Used as part of integrated programs (seed treatment + soil drench + foliar) to boost overall plant vigor and reduce chemical input needs. 2. Improving Soil Health and Residue Breakdown 2.1 Faster composting and bokashi-style fermentation Lactic acid bacteria like L. plantarum are excellent at fermenting organic materials. seedandknowledge+1 On-farm examples Bokashi compost: Farmers and gardeners mix L. plantarum–rich inoculants with kitchen scraps, crop residues, or manures in airtight containers. The material ferments instead of rotting, then is buried or added to soil, where it finishes breaking down quickly. Compost accelerators: LAB-based “starters” are sprayed on compost piles to speed up breakdown and reduce odors. Benefits Faster conversion of waste into usable organic fertilizer. Less smell and fewer flies around manure or food waste piles. More stable, nutrient-rich organic matter that feeds soil life. pmc.ncbi.nlm.nih+1 2.2 Supporting soil structure and nutrient cycling By helping break down organic matter and producing sticky substances and acids, L. plantarum contributes indirectly to: Better soil aggregation (crumb structure), which improves water infiltration and root penetration. Smoother nutrient cycling, so nitrogen, phosphorus, and other elements are released in plant-friendly forms. publishing.emanresearch+1 A simple way to picture it: L. plantarum and other LAB act like a “starter culture” for your soil, waking up the biology that drives healthy nutrient flows. 3. Natural Support Against Plant Diseases 3.1 Suppressing harmful fungi and bacteria L. plantarum produces lactic acid, antimicrobial peptides (bacteriocins), and sometimes biosurfactants that can slow down or block certain pathogens. frontiersin+2 Research on lactic acid bacteria in agriculture shows they can: Reduce fungal diseases like Fusarium on cereals and horticultural crops. Inhibit spoilage and disease organisms on fruits and vegetables. pmc.ncbi.nlm.nih+1 Practical examples Seed or root dips: Seeds or seedlings dipped in a suspension containing L. plantarum before planting to protect them from early soil-borne infections. Foliar sprays (in some programs): LAB-based sprays used alongside other biologicals to lower disease pressure on leaves and fruit surfaces. While L. plantarum alone is not a full replacement for all fungicides, it adds a valuable biological layer of protection in integrated pest management. 3.2 Better silage and forage preservation In forage production, L. plantarum is widely used as a silage inoculant. Studies on soybean and amaranth silage show that adding L. plantarum: Lowers pH faster. Increases lactic acid content. Improves protein preservation and overall fermentation quality. frontiersin+1 On-farm outcome Dairy and livestock producers get: More stable silage with less spoilage. Better nutritional value, which supports animal performance and reduces feed losses. 4. Uses in Animal Nutrition and Health 4.1 As a probiotic or postbiotic in feed L. plantarum is a well-established probiotic in animal nutrition.[ frontiersin ] In poultry and livestock Postbiotics (heat-killed cells and their metabolites from L. plantarum) have been tested as alternatives to antibiotic growth promoters in broiler chickens. Results show improved gut health, better nutrient digestibility, stronger immune response, and improved growth and meat quality.[ frontiersin ] In ruminants and pigs L. plantarum from silage and supplements supports a balanced gut microbiota, helping animals use feed more efficiently and resist digestive upsets. frontiersin+1 This use connects back to soil: healthier animals mean better-quality manure, which returns to fields and feeds soil microbes, including LAB. 5. Uses in Food, Fermentation, and Bioproducts Although your main interest may be agriculture and soil, many commercial uses of L. plantarum come from food and biotech, and they often tie back to farming systems. 5.1 Food fermentation and shelf-life extension L. plantarum is widely used as: A starter culture for fermented vegetables, dairy, cereals, meats, and plant-based drinks. pmc.ncbi.nlm.nih+1 A bio-preservative to keep food safer for longer by suppressing spoilage organisms and pathogens. frontiersin+1 This matters for agriculture because it: Creates more stable markets for crops (e.g., vegetables and grains processed into high-value fermented products). Reduces food losses post-harvest. 5.2 Human probiotic products In the human health sector, L. plantarum is used in probiotic capsules, drinks, and yogurts to support digestion, immunity, and general well-being. frontiersin+1 While this is outside the field, it reinforces one key point: the same microbe that supports human gut health can also support “soil gut health” when applied correctly in agriculture. 5.3 Industrial enzymes and biosurfactants Certain strains of L. plantarum are used industrially to produce: Enzymes (amylases, glucosidases, lipases) for food processing and other applications.[ pmc.ncbi.nlm.nih ] Biosurfactants with strong antimicrobial and antibiofilm activity that can be used in cleaning, coatings, and possibly crop protection formulations. frontiersin+1 These higher-value products can originate from agricultural feedstocks (grains, molasses, plant juices), creating new markets for farm outputs. 6. Emerging Uses: Biofertilizer R&D and Functional Crops New research and projects are exploring more targeted uses of lactic acid bacteria like L. plantarum as plant inoculants: EU-backed work on LAB-based biofertilizers for blueberries aims to increase yield and boost health-promoting compounds in the fruit, all while keeping biosafety standards high.[ cordis.europa ] Reviews highlight LAB as promising tools for pesticide detoxification, heavy-metal bioremediation, and more advanced soil restoration strategies. publishing.emanresearch+1 As these technologies mature, farmers may see more crop-specific products where L. plantarum is selected not just for general soil health, but for specific benefits like improving fruit antioxidant content or helping plants cope with stress. 7. Bringing It All Together Across agriculture, soil systems, and related industries, the main practical uses of Lactiplantibacillus plantarum are: In the soil: speeding up organic matter breakdown, improving nutrient availability, and supporting a healthy soil microbiome. In biofertilizers: acting as a core member of microbial blends that revitalize soils, promote root growth, and reduce reliance on chemicals. In plant protection: adding a natural layer of disease suppression against harmful fungi and bacteria, and improving silage preservation. In animals and food: serving as a probiotic, postbiotic, and starter culture that links farm production to safe, high-value feed and food. frontiersin+2
- Lactiplantibacillus Plantarum: Benefits, Functions, and Characteristics Across Industries
Photo credit: https://www.vital.ly/trc/Lactiplantibacillus-plantarum/monograph=1548/?srsltid=AfmBOoqoZblaCDSoP4cwnLRoLqhfYFU-Fdif66WK81yCI6u0nV49KzQ0 Lactiplantibacillus Plantarum (historically known as Lactobacillus plantarum) is a lactic acid bacterium best known from fermented foods and probiotics, but it is also emerging as a powerful tool in agriculture. Its ability to ferment organic matter, produce organic acids and antimicrobial compounds, and adapt to diverse environments makes it a versatile component of modern biofertilizers and soil microbial blends. indogulfbioag+1 This overview explains, in simple and practical terms, how L. plantarum behaves at the microbial level, how it benefits soil and crops, and where it fits into industry-scale biofertilizer and soil health programs. 1. What Is Lactiplantibacillus Plantarum? L. plantarum is a Gram-positive, facultative heterofermentative lactic acid bacterium. In practice, that means: It ferments many different plant sugars into lactic acid as the main product, and can also produce acetic acid and other metabolites depending on conditions.[ pmc.ncbi.nlm.nih ] It tolerates both low pH and moderate oxygen, so it can survive in compost heaps, biofertilizer tanks, rhizosphere soils, plant residues, and even the animal gut. emnz+1 It has a relatively large and flexible genome for a lactic acid bacterium, with many genes for carbohydrate transport and metabolism, stress tolerance, and antimicrobial compound production. frontiersin+1 Genomic studies show that L. plantarum strains often carry genes for multiple bacteriocins (e.g., plantaricin E and F, Enterocin X) and other secondary metabolites with antimicrobial activity, giving them a strong competitive advantage in mixed microbial environments. frontiersin+1 These traits make L. plantarum a “generalist” microbe that adapts well from food systems to soil and plant systems. 2. Core Microbial Functions in Soil and Rhizosphere 2.1 Fermentation of Organic Matter In soil and organic amendments, L. plantarum: Ferments carbohydrates from crop residues, manures, and plant exudates into lactic acid and other organic acids. Drives a controlled “mini-fermentation” of organic matter, similar to silage or fermented foods, but now taking place in soil or compost piles. frontiersin+1 This fermentation: Speeds up decomposition and humus formation. Reduces foul odors and ammonia emissions from manures and immature composts. pmc.ncbi.nlm.nih+1 Helps stabilize organic matter so nutrients are released more gradually. 2.2 Local pH Shifts and Nutrient Solubilization Lactic acid and other organic acids from LAB (lactic acid bacteria) locally lower pH in the micro-zone around decomposing residues or root surfaces. This has several soil benefits: Phosphorus solubilization: LAB can convert insoluble phosphate minerals into plant-available forms in phosphate-accumulated or saline soils. emnz+1 Micronutrient availability: Acidification and chelating metabolites increase the solubility of iron, zinc, and other micronutrients, helping crops in high-pH or compacted soils. publishing.emanresearch+1 Reviews on LAB in sustainable agriculture consistently report that these bacteria improve soil structure and fertility by accelerating organic matter breakdown, solubilizing phosphorus, and balancing microbial communities. pmc.ncbi.nlm.nih+1 2.3 Biocontrol and Pathogen Suppression L. plantarum protects plants and soil systems through several mechanisms: Acidification: Many fungal and bacterial pathogens are less competitive in low-pH micro-environments created by lactic acid. emnz+1 Bacteriocins and antifungal metabolites: Strains produce bacteriocins (e.g., plantaricins) and antifungal compounds such as phenyllactic acid and related phenolic acids that inhibit molds like Aspergillus, Penicillium, and Fusarium. sciencedirect+2 In cereals, L. plantarum and related LAB have been used to reduce Fusarium head blight and associated mycotoxins. publishing.emanresearch+1 Competition and biofilms: Some strains form biofilms on root surfaces, occupying space and using nutrients so pathogens find it harder to establish. agritechinsights+1 A detailed functional study of 25 L. plantarum strains showed that several isolates strongly inhibited toxigenic fungi and also stimulated cereal germination and growth, linking antifungal activity with plant-beneficial effects.[ sciencedirect ] 3. Plant Growth Promotion: Evidence from Field and Greenhouse Studies 3.1 Seed Germination and Seedling Vigor Several studies have tested L. plantarum directly on seeds: Wheat seeds treated with individual or mixed L. plantarum strains showed: 6–40% higher germination (depending on conditions and inoculum level). Seedling height increases of 8–41%. Root length increases up to 2.4-fold in hydroponics and 6.8–64.5% in soil.[ agris.fao ] Microscopy in the same study showed that mixed L. plantarum cultures formed biofilms on wheat roots, explaining the strong root growth response.[ agris.fao ] 3.2 Rhizosphere Colonization and Growth Promotion A recent tomato study on a soil–plant system found that a specific L. plantarum strain (LP0308): Stably colonized the rhizosphere over at least 20 days. Increased plant height, bud length, primary root length, and root and seedling fresh weight. Shifted the soil microbial community, increasing beneficial Bacillus spp. and reducing pathogens like Ralstonia solanacearum and Fusarium oxysporum.[ agritechinsights ] This type of “microbiome engineering” is important: L. plantarum is not acting alone, but reshaping the surrounding community toward a more suppressive and nutrient-efficient soil. 3.3 Tolerance to Drought and Heat Stress Work in wheat under combined drought and heat stress has shown that L. plantarum and related Lactobacillus strains: Increase chlorophyll a and b and carotenoid levels under stress, supporting photosynthesis. Enhance antioxidant enzyme activities (catalase, peroxidase, superoxide dismutase, ascorbate peroxidase), which reduce oxidative damage in stressed plants.[ jksus ] These responses translate into better growth and yield stability under adverse conditions, which is particularly valuable in semi-arid and climate-stressed regions. 4. Roles in Biofertilizers and Soil Microbial Blends 4.1 As a Stand‑Alone Microbial Species for Formulators L. plantarum is now offered as a defined microbial species for custom formulations, typically at strengths of 1 × 10^8 to 1 × 10^9 CFU per gram.[ indogulfbioag ] Core marketed benefits include: Acting as a “probiotic” in the rhizosphere: enhancing root development and nutrient uptake. Supporting organic matter breakdown and fermentation-based soil improvement. Contributing antimicrobial and competitive functions in multi-strain products.[ indogulfbioag ] Because it is widely studied in food and health contexts and generally recognized as safe at strain level, regulators and industry often view L. plantarum as a low-risk but high-impact candidate for biofertilizer design. pmc.ncbi.nlm.nih+1 4.2 As Part of Effective Microorganisms (EM)-Type Blends Many commercial “Effective Microorganisms” or microbial blends use L. plantarum as one of several core species. Typical consortia combine: Lactic acid bacteria (including L. plantarum). Photosynthetic bacteria such as Rhodopseudomonas palustris. Yeasts such as Saccharomyces cerevisiae. Sometimes Bacillus and Bifidobacterium species, and in some products arbuscular mycorrhizal fungi. indogulfbioag+2 Examples from IndoGulf BioAg’s portfolio: Microm – an EM-type blend where L. plantarum is one of several organisms at 1 × 10^8 CFU/ml; marketed to improve soil fertility and plant growth by promoting fermentation and beneficial microbial environments.[ indogulfbioag ] Micro-Manna – a microbial activator that contains lactic acid bacteria including L. plantarum along with Bacillus and Bifidobacterium, designed to enhance performance of biofertilizers and favor beneficial soil microbes.[ indogulfbioag ] BoostX – a crop kit blend listing L. plantarum among multiple Lactobacillus, Bifidobacterium, yeast and photosynthetic bacteria, used to influence the microbial environment around roots and support plant growth and soil fertility.[ indogulfbioag ] In these products, L. plantarum’s role is to: Start fermentation quickly by rapidly converting available sugars to lactic acid. Create low-pH microzones that suppress opportunistic pathogens. Pre-digest organic inputs, making them more accessible to other beneficial microbes and plant roots. pmc.ncbi.nlm.nih+1 4.3 In Composting, Bokashi, and On‑Farm Ferments Farm-scale practices such as bokashi composting and fermented plant/food wastes rely heavily on lactic acid bacteria: LAB inoculants speed up the breakdown of lignin- and cellulose-rich residues. Fermented material becomes richer in stabilized organic matter and plant-available nutrients. Ammonia and odor emissions drop significantly as LAB convert nitrogen forms and trap them in microbial biomass and organic complexes. publishing.emanresearch+2 L. plantarum is often a dominant LAB in such systems because of its wide substrate range and stress tolerance. 5. Interactions with Soil Microbiome and Nutrient Cycles 5.1 Supporting Soil Biological Structure LAB-based biofertilizers help rebuild degraded soil biology by: Improving soil aggregation and porosity via exopolysaccharides and biofilms, leading to better aeration and water infiltration. emnz+1 Increasing microbial diversity and functional redundancy, which is key for long-term disease suppression and nutrient cycling. pmc.ncbi.nlm.nih+1 Creating niches that favor beneficial groups such as Bacillus, actinobacteria, and mycorrhizal fungi, particularly when applied with organic amendments. linkedin+1 5.2 Integration with Nitrogen and Phosphorus Cycles L. plantarum does not fix nitrogen, but it shapes N and P availability indirectly: Enhances decomposition of manures and residues, releasing organic nitrogen in more plant-available forms. Solubilizes phosphate from insoluble pools, complementing phosphorus-solubilizing bacteria and mycorrhizae. publishing.emanresearch+1 Helps mitigate the negative effects of long-term mineral fertilizer overuse by supporting a richer, more balanced soil microbiome that can restore functions like disease suppression and nutrient cycling. frontiersin+1 When combined with nitrogen-fixing bacteria and P-solubilizers, LAB-based products allow gradual reduction of synthetic NPK rates while maintaining yields, as shown in broader microbial biofertilizer studies. linkedin+1 6. How L. plantarum Survives and Performs at Microbial Level From a microbiology and formulation perspective, several characteristics explain why L. plantarum is attractive for industry: Environmental tolerance: Many strains grow well between pH ~3–7 and survive mild salinity and temperature fluctuations, important for storage and field application.[ pmc.ncbi.nlm.nih ] Metabolic flexibility: The genome encodes numerous sugar transporters and metabolic pathways, enabling growth on diverse plant sugars in soil, compost, and root exudates. frontiersin+1 Antimicrobial arsenal: Bacteriocins (plantaricins and others) targeting closely related bacteria, including some pathogens. Organic acids, hydrogen peroxide, and phenolic compounds that inhibit fungi and Gram-negative bacteria. frontiersin+2 Biofilm formation: Surface-associated growth on roots or organic particles protects cells against environmental stress and allows long-term colonization. agritechinsights+1 These features mean that, once introduced via a biofertilizer or EM-type product, L. plantarum can persist long enough to influence the rhizosphere and residue decomposition, even if it does not become a dominant permanent member of the soil community. 7. Practical Considerations for Growers and Industry 7.1 When to Use L. plantarum–Based Products L. plantarum–containing biofertilizers are especially useful when: Soils have high organic residues but poor biological activity (post-intensive fertilizer use, low organic inputs). There is a history of soil-borne disease pressure (Fusarium, Rhizoctonia, Pythium) and a need for biological suppression. Fields are saline, compacted, or suffering from nutrient lockup; LAB can help solubilize bound phosphorus and improve organic matter turnover. linkedin+2 Systems are transitioning to organic or reduced-chemical regimes and need a biological “kick-start” for the soil microbiome. 7.2 Application and Compatibility Key operational points: Dose and frequency: Typical EM-type soil applications are in the range of 10^7–10^8 CFU/ml formulations, applied as soil drenches, drip injections, or mixed with compost teas, often every 2–4 weeks depending on product guidelines. indogulfbioag+1 Carriers: Liquid concentrates allow easy mixing with irrigation but need careful storage (cool, out of direct sun). Dry carriers (powders, granules) offer longer shelf life but must be rehydrated properly. Compatibility: Avoid tank-mixing with alkaline solutions, strong oxidants, or high-copper fungicides, which can kill LAB. Compatible with most organic fertilizers, compost extracts, and other microbial inoculants when applied as separate passes or with appropriate pH control. 7.3 Integration into Broader Biological Programs Best results come when L. plantarum is integrated into a broader program rather than used in isolation: Combine with Bacillus (for nitrogen cycling, P and K solubilization, and strong antibiosis) and Trichoderma or mycorrhizal fungi (for root protection and nutrient uptake) in well-designed consortia. linkedin+1 Use alongside organic matter inputs (composts, green manures) so LAB has substrates to ferment and transform. Adjust mineral fertilizer rates gradually as soil biological indicators and crop performance improve, to avoid yield shocks. 8. Summary Lactiplantibacillus plantarum is far more than a food probiotic. In agricultural and soil systems, it: Ferments organic matter, stabilizes residues, and improves soil structure. Solubilizes phosphorus and enhances nutrient availability through localized acidification and metabolite production. Suppresses pathogens via organic acids, bacteriocins, and antifungal metabolites. Promotes seed germination, root growth, and stress tolerance, especially when used as part of multi-strain biofertilizers. sciencedirect+3 Integrates well into EM-style blends and comprehensive soil health programs that aim to reduce chemical inputs and restore biological function. indogulfbioag+3 For biofertilizer manufacturers , agronomists, and progressive growers, L. plantarum offers a robust, scientifically supported component for next-generation microbial products—bridging food microbiology, soil ecology, and practical crop production in a single, highly adaptable species.
- How Does Corynebacterium Spp. Improve Plant Immunity?
Corynebacterium spp ., a group of beneficial soil bacteria, enhances plant immunity by solubilizing manganese—a key micronutrient for defense enzymes—and triggering systemic resistance pathways against pathogens and stresses. As a plant growth-promoting rhizobacterium (PGPR), it improves nutrient uptake, root health, and overall resilience, making it valuable for sustainable agriculture. indogulfbioag+1 What Are Corynebacterium spp. role in Agriculture? Corynebacterium species are Gram-positive, rod-shaped bacteria naturally found in soils, often part of manganese-solubilizing consortia alongside fungi like Penicillium citrinum. In bioagriculture, select strains (1 x 10⁸-10⁹ CFU/g) colonize plant roots, converting insoluble soil manganese (MnO₂) into plant-available Mn²⁺ via organic acids (citric, gluconic, oxalic) and chelation.[ indogulfbioag ][ ppl-ai-file-upload.s3.amazonaws ] This solubilization supports photosynthesis, enzyme activation, and oxidative stress defense, directly linking to stronger plant immunity. Recommended for cereals, vegetables, fruits, and ornamentals, it's compatible with biofertilizers but not chemicals.[ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] How Manganese Solubilization Boosts Plant Defenses Essential Role of Manganese in Immunity Manganese activates superoxide dismutase (Mn-SOD), a frontline antioxidant enzyme neutralizing reactive oxygen species (ROS) during pathogen attacks or drought. Corynebacterium spp. ensure Mn availability in deficient soils (alkaline or sandy), preventing chlorosis and weak defenses.[ indogulfbioag ] Plants with ample Mn produce more lignin—a physical barrier against fungi—and phenolics for antimicrobial action. Field applications show treated crops exhibit 20-30% better stress tolerance via enhanced Mn-SOD activity.[ indogulfbioag ] Organic Acid Production Mechanism Bacteria lower rhizosphere pH, dissolving Mn oxides: organic acids chelate Mn²⁺ for root uptake. This indirect immunity boost reduces oxidative damage, priming plants for biotic challenges like Fusarium or Rhizoctonia. indogulfbioag+1 Triggering Induced Systemic Resistance (ISR) Molecular Signaling Pathways Corynebacterium spp., like other PGPR (Pseudomonas, Bacillus), elicit ISR via jasmonic acid (JA) and ethylene (ET) pathways, distinct from salicylic acid (SA)-based SAR. Root colonization releases lipopolysaccharides or siderophores, activating MYB72 transcription factor for systemic priming. annualreviews+3 ISR upregulates PDF1.2 (JA/ET marker) and WRKY genes, enhancing defenses against necrotrophs and insects without constant energy cost—plants "remember" for faster response. pmc.ncbi.nlm.nih+1 Evidence from PGPR Studies While Corynebacterium-specific ISR data is emerging, analogous strains show 40-60% disease reduction (e.g., against Pythium). Combined with Mn supply, it synergizes ISR, as Mn supports JA biosynthesis enzymes. indogulfbioag+2 Enhancing Physical and Biochemical Barriers Root Development and Soil Aeration Corynebacterium promotes root elongation and branching, increasing nutrient absorption and exudate release that recruits more beneficial microbes. Improved aeration reduces anaerobic pathogens.[ pmc.ncbi.nlm.nih ][ ppl-ai-file-upload.s3.amazonaws ] Lignin deposition in roots, Mn-activated, fortifies against nematodes and root rots.[ indogulfbioag ] Antimicrobial Compounds and Competition Strains produce siderophores, starving pathogens of iron, and enzymes degrading fungal cell walls. This biocontrol complements immunity, reducing disease incidence by 30-50% in pulses and oilseeds.[ indogulfbioag ][ ppl-ai-file-upload.s3.amazonaws ] Stress Tolerance: Drought and Salinity Immunity Abiotic Stress Crosstalk ISR from Corynebacterium cross-protects against drought via osmolyte (proline, glycine betaine) accumulation and stomatal regulation. Mn-SOD quenches drought-induced ROS, maintaining photosynthesis. nature+2 Inoculated cactus pear showed higher N accumulation and metabolism under water scarcity. Salinity trials reveal better ionic balance, as Mn aids Na⁺ exclusion. indogulfbioag+1 Stress Tolerance Comparison Table: Stress Type Corynebacterium Effect Key Mechanism indogulfbioag+1 Drought 25-40% yield protection Mn-SOD, ISR (JA/ET) Salinity Improved water uptake Osmolytes, root growth Oxidative Reduced ROS damage Antioxidant activation Crop-Specific Immunity Benefits Cereals and Pulses Seed treatment (10-15g/kg) boosts wheat/barley immunity to rusts via Mn-enhanced phenolics; 15-20% yield gain reported.[ ppl-ai-file-upload.s3.amazonaws ] Vegetables and Fruits Soil drench protects tomatoes from wilt, activating PR genes indirectly through ISR-like priming.[ indogulfbioag ] Plantation Crops Orchards see better anthracnose resistance from root health and Mn nutrition.[ ppl-ai-file-upload.s3.amazonaws ] Application Methods for Maximum Immunity Seed Coating: 10-15g/kg seeds; shade-dry.[ ppl-ai-file-upload.s3.amazonaws ] Seedling Dip: 100g/ sufficient water, 30min soak.[ ppl-ai-file-upload.s3.amazonaws ] Soil Incorporation: 2.5-5kg/ha with manure.[ ppl-ai-file-upload.s3.amazonaws ] Drip Irrigation: 2.5-5kg/ha solution.[ ppl-ai-file-upload.s3.amazonaws ] Shelf-stable 1 year; apply pre-planting for colonization.[ ppl-ai-file-upload.s3.amazonaws ] Scientific Backing and Future Potential Studies on PGPR confirm ISR efficacy; Corynebacterium's Mn role uniquely ties nutrition to immunity. Genomics reveal WRKY/MYB regulation, promising for engineered strains. pmc.ncbi.nlm.nih+3 In sustainable farming, it cuts chemical use by 30%, promoting microbiome diversity.[ indogulfbioag ] For detailed FAQs on Corynebacterium spp., including dosage and compatibility, visit: https://www.indogulfbioag.com/microbial-species/corynebacterium-spp. [ ppl-ai-file-upload.s3.amazonaws ]
- What is the Difference Between BTI and Chemical Larvicides?
Bacillus thuringiensis israelensis (Bti) stands out as a biological larvicide that selectively targets mosquito and black fly larvae, differing markedly from chemical larvicides in safety, environmental persistence, and mode of action. Chemical larvicides like temephos or methoprene offer broad-spectrum control but carry risks of resistance and non-target harm that Bti largely avoids. fpls-11-00071.pdf+1 epa+2 Core Differences between BTI and chemical larvicides: Composition and Mode of Action Bti is a naturally occurring soil bacterium that, during sporulation, produces delta-endotoxins (Cry and Cyt proteins) packaged in parasporal crystals. When mosquito larvae ingest these crystals in water, the alkaline gut solubilizes them into protoxins, which bind specific receptors on gut epithelial cells, form membrane pores, and disrupt the gut barrier. This leads to paralysis, bacterial proliferation in the hemocoel, and death within 24-72 hours, without affecting non-susceptible organisms. fmicb-14-1293302.pdf+1 pmc.ncbi.nlm.nih+1 Chemical larvicides, conversely, are synthetic compounds: organophosphates (e.g., temephos) inhibit acetylcholinesterase enzymes, carbamates (e.g., bendiocarb) reversibly block the same, insect growth regulators (IGRs) like methoprene mimic juvenile hormones to disrupt metamorphosis, and pyrethroids (e.g., permethrin) alter sodium channels in nerves. These act systemically or on contact, impacting a wide range of insects and sometimes vertebrates. nitrogenbacteria.wixsite+3 This biological vs. synthetic divide means Bti requires ingestion and specific gut conditions, providing inherent specificity, while chemicals diffuse broadly in water.[ pmc.ncbi.nlm.nih ][ ppl-ai-file-upload.s3.amazonaws ] Target Specificity and Spectrum Bti's Narrow Focus Bti primarily affects larvae of Diptera like mosquitoes (Aedes, Culex, Anopheles), black flies (Simulium), and some midges, due to unique receptor proteins in their guts. It spares beneficial insects (bees, ladybugs), aquatic predators (dragonfly nymphs), crustaceans (Daphnia), fish, amphibians, and mammals. nature+2 fpls-11-00071.pdf+1 This precision suits sensitive ecosystems like wetlands or organic farms.[ epa ][ ppl-ai-file-upload.s3.amazonaws ] Chemical Broad Reach Chemicals kill indiscriminately: temephos targets cholinesterase in many arthropods, methoprene affects aquatic invertebrates broadly, and pyrethroids harm fish and amphibians via gill absorption. Non-target mortality disrupts food webs, reducing natural mosquito predators. pmc.ncbi.nlm.nih+3 Efficacy Profiles Speed and Duration Bti kills 80-100% of larvae in 1-3 days under optimal conditions, with briquettes providing 14-30 days in contained water. Efficacy holds in moderately polluted water but dips in highly organic sites unless combined with surfactants. pmc.ncbi.nlm.nih+1 [ ppl-ai-file-upload.s3.amazonaws ] Chemicals act faster (hours for organophosphates), with residuals up to 4 weeks for IGRs, but performance varies with pH, turbidity, and organic load. pmc.ncbi.nlm.nih+1 Metric Bti Chemical Larvicides Time to Mortality 24-72 hours [ ppl-ai-file-upload.s3.amazonaws ][ nature ] Hours-24 hours [ pmc.ncbi.nlm.nih ] Residual Control 7-30 days [ ppl-ai-file-upload.s3.amazonaws ] 7-60 days [ pmc.ncbi.nlm.nih ] Water Tolerance Good in low-moderate organics [ nature ] Variable; adsorbs to organics [ pmc.ncbi.nlm.nih ] Bti excels in repeated, low-dose use.[ nitrogenbacteria.wixsite ] Safety for Humans, Animals, and Applicators Bti's Clean Record No acute or chronic toxicity to humans; EPA Reduced Risk status confirms safety for drinking water application. Pets, livestock, and wildlife unaffected; no bioaccumulation. Applicators need basic PPE for dusts. indogulfbioag+1 fpls-11-00071.pdf+1 Chemical Risks Organophosphates cause cholinergic poisoning (sweating, vomiting); IGRs have low acute risk but potential endocrine disruption; pyrethroids irritate skin/respiratory. Fish LC50 often <1 ppb; bird/mammal risks higher. sciencedirect+2 Safety Factor Bti Chemical Larvicides Human LD50 >5,000 mg/kg [ epa ] 50-2,000 mg/kg [ pmc.ncbi.nlm.nih ] Fish Toxicity None [ ppl-ai-file-upload.s3.amazonaws ][ pmc.ncbi.nlm.nih ] High (ppb levels) [ pmc.ncbi.nlm.nih ] Applicator Risk Low [ ppl-ai-file-upload.s3.amazonaws ] Moderate-high [ pmc.ncbi.nlm.nih ] Bti enables community programs without evacuation.[ indogulfbioag ] Environmental Fate and Persistence Bti Degradation UV light, dilution, and microbes inactivate Bti within 2-14 days; spores persist but non-pathogenic. No groundwater contamination or biomagnification. indogulfbioag+1 fpls-11-00071.pdf+1 Chemical Persistence Temephos half-life 1-7 days but forms toxic oxons; methoprene 10-20 days; pyrethroids bind sediments, releasing slowly. Runoff pollutes distant waters. pmc.ncbi.nlm.nih+1 Environmental Trait Bti Chemical Larvicides Half-Life Days [ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] Days-weeks [ pmc.ncbi.nlm.nih ] Runoff Risk Negligible [ ppl-ai-file-upload.s3.amazonaws ] High [ pmc.ncbi.nlm.nih ] Bioaccumulation None [ pmc.ncbi.nlm.nih ] Possible [ pmc.ncbi.nlm.nih ] Bti supports biodiversity. pmc.ncbi.nlm.nih+1 Resistance Management Bti's four synergistic toxins bind multiple sites, yielding no field resistance after 40+ years; lab strains show <10-fold shifts. Rotate with L. sphaericus.[ ppl-ai-file-upload.s3.amazonaws ] nature+1 Chemicals face multi-fold resistance globally (e.g., Aedes to temephos); treadmill effect demands new molecules. pmc.ncbi.nlm.nih+1 Resistance Status Bti Chemical Larvicides Field Cases None significant [ nature ] Widespread [ pmc.ncbi.nlm.nih ] Fold Increase Low (<10x) [ ppl-ai-file-upload.s3.amazonaws ] High (100-1000x) [ pmc.ncbi.nlm.nih ] Bti preserves efficacy.[ nitrogenbacteria.wixsite ] Cost, Regulations, and Practical Use Economic and Operational Aspects Bti costs $0.01-0.05/m², comparable initially but cheaper long-term sans resistance management. Granules/briquettes simplify application in hard-to-reach sites; aerial feasible. pmc.ncbi.nlm.nih+1 [ ppl-ai-file-upload.s3.amazonaws ] Chemicals cheaper ($0.005-0.03/m²) but incur monitoring, disposal, and buffer zone costs.[ nitrogenbacteria.wixsite ] Regulations favor Bti: WHO-recommended, organic-approved; chemicals face restrictions/re-registration.[ ppl-ai-file-upload.s3.amazonaws ][ epa ] Application Scenarios Scenario Preferred Choice Reason Urban Catch Basins Bti [ indogulfbioag ] Safety near homes Large Wetlands Bti [ pmc.ncbi.nlm.nih ] Ecology protection Emergency Outbreak Chemicals [ pmc.ncbi.nlm.nih ] Speed Organic Farms Bti [ ppl-ai-file-upload.s3.amazonaws ] Certification Integration in Modern Programs Bti fits IVM as a rotation partner, reducing chemical reliance by 70-90% in many regions. Hybrids combine strengths. pmc.ncbi.nlm.nih+1 For comprehensive FAQs on Bti mosquito control safety, check: https://www.indogulfbioag.com/post/bti-mosquito-control-safety [ ppl-ai-file-upload.s3.amazonaws ]
- How Does Bti Compare to Chemical Pesticides?
Bacillus thuringiensis israelensis (Bti) offers a targeted, biological alternative to chemical pesticides for mosquito and black fly control, primarily by killing larvae in water without broad environmental harm. While chemical pesticides like organophosphates, carbamates, and pyrethroids provide fast knockdown, Bti excels in safety, sustainability, and resistance management. indogulfbioag+1 Key Comparison Areas Bti and chemical pesticides differ fundamentally in mode of action, spectrum, and long-term implications. Bti produces crystal proteins that larvae ingest and activate in their alkaline gut, forming pores that cause septicemia within 24-48 hours. Chemicals, by contrast, often act via nerve disruption or respiration inhibition, affecting adults and larvae alike across many species. fmicb-14-1293302.pdf+1 nitrogenbacteria.wixsite+1 This specificity makes Bti ideal for integrated vector management, while chemicals risk killing beneficial insects and building resistance. nitrogenbacteria.wixsite+1 [ ppl-ai-file-upload.s3.amazonaws ] Efficacy and Speed Bti Performance Bti achieves 90-100% larval mortality in field trials within days, especially against Aedes, Culex, and Anopheles species. It works best on early instars in clean to moderately polluted water, with formulations like granules or briquettes providing 7-14 days of control. indogulfbioag+2 [ ppl-ai-file-upload.s3.amazonaws ] Programs in the US, Canada, and Europe report sustained reductions in adult mosquitoes after repeated applications. indogulfbioag+1 Chemical Performance Chemicals such as temephos (organophosphate) or methoprene (insect growth regulator) offer quicker results, often killing larvae in hours, and some have longer residuals up to 30 days. However, efficacy drops in organic-rich water due to binding or degradation. pmc.ncbi.nlm.nih+1 In high-density outbreaks, chemicals provide immediate knockdown but require frequent reapplication due to resistance. pmc.ncbi.nlm.nih+1 Direct Comparison Aspect Bti Chemical Pesticides Larval Kill Time 24-48 hours [ ppl-ai-file-upload.s3.amazonaws ][ nature ] Hours to 1 day [ pmc.ncbi.nlm.nih ] Residual Effect 7-14 days [ ppl-ai-file-upload.s3.amazonaws ][ pmc.ncbi.nlm.nih ] 7-30 days [ pmc.ncbi.nlm.nih ] Field Efficacy High in targeted habitats indogulfbioag+1 High but declines with resistance [ pmc.ncbi.nlm.nih ] Reapplication Frequency Weekly in rain [ ppl-ai-file-upload.s3.amazonaws ] Less frequent but environmental buildup [ nitrogenbacteria.wixsite ] Bti matches chemicals in controlled settings but shines in sustainable programs. nature+1 Safety for Humans and Animals Bti Safety The EPA classifies Bti as non-toxic to humans, with no risks from ingestion, inhalation, or skin contact at labeled rates. It poses no threat to pets, livestock, fish, birds, or amphibians, even in drinking water. epa+1 fpls-11-00071.pdf+1 Mild irritation from concentrates is rare and mitigated by PPE.[ ppl-ai-file-upload.s3.amazonaws ] Chemical Safety Organophosphates like malathion inhibit acetylcholinesterase, causing nausea, dizziness, or worse in humans; pyrethroids irritate skin and eyes. Acute poisonings occur in applicators and communities, with chronic links to neurological issues. pmc.ncbi.nlm.nih+1 Pets and wildlife suffer similarly, with fish highly sensitive to many larvicides.[ pmc.ncbi.nlm.nih ] Direct Comparison Aspect Bti Chemical Pesticides Human Toxicity None [ ppl-ai-file-upload.s3.amazonaws ][ epa ] Moderate to high [ pmc.ncbi.nlm.nih ] Pet/Livestock Safe [ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] Risky, especially aquatics [ pmc.ncbi.nlm.nih ] Application PPE Minimal [ ppl-ai-file-upload.s3.amazonaws ] Full protective gear [ pmc.ncbi.nlm.nih ] Bti's profile supports residential and organic use.[ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] Environmental Impact Bti Impact Bti biodegrades in days to weeks via sunlight and microbes, leaving no residues. It spares bees, predators, and most aquatic life due to gut-specific action; minor indirect effects on chironomids occur only under heavy use. sciencedirect+2 fpls-11-00071.pdf+1 Approved for wetlands and organics.[ epa ][ ppl-ai-file-upload.s3.amazonaws ] Chemical Impact Chemicals persist longer, contaminating soil, water, and food chains; they kill non-target invertebrates, fish, and birds. Runoff causes algal blooms and biodiversity loss. nitrogenbacteria.wixsite+1 Pyrethroids harm aquatic organisms at low levels.[ pmc.ncbi.nlm.nih ] Direct Comparison Aspect Bti Chemical Pesticides Biodegradation Rapid (days-weeks) [ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] Variable, often persistent [ pmc.ncbi.nlm.nih ] Non-Target Effects Minimal [ ppl-ai-file-upload.s3.amazonaws ][ pmc.ncbi.nlm.nih ] Broad (bees, fish, etc.) nitrogenbacteria.wixsite+1 Water/Soil Residue None [ ppl-ai-file-upload.s3.amazonaws ] Accumulates [ pmc.ncbi.nlm.nih ] Organic Approval Yes [ ppl-ai-file-upload.s3.amazonaws ] Rarely [ indogulfbioag ] Bti preserves ecosystems better. nature+1 Resistance Development Bti Resistance Multi-toxin strategy (Cry4, Cry11, Cyt1) prevents widespread resistance; no field cases after decades, only lab-selected low-level shifts. Rotate with B. sphaericus for longevity. indogulfbioag+1 [ ppl-ai-file-upload.s3.amazonaws ] Chemical Resistance Mosquitoes rapidly evolve resistance to single-site chemicals like pyrethroids (now widespread globally). "Pesticide treadmill" requires escalating doses or switches. pmc.ncbi.nlm.nih+1 Direct Comparison Aspect Bti Chemical Pesticides Resistance Risk Very low [ ppl-ai-file-upload.s3.amazonaws ][ nature ] High, common pmc.ncbi.nlm.nih+1 Management Rotation easy [ indogulfbioag ] Frequent new chemistries needed [ pmc.ncbi.nlm.nih ] Long-Term Viability High (40+ years) [ indogulfbioag ] Declining [ pmc.ncbi.nlm.nih ] Bti sustains control longer.[ nitrogenbacteria.wixsite ] Cost and Practicality Bti Practicality Initial costs match chemicals, but lower reapplication and no cleanup reduce totals; easy formulations for ground/aerial use. Consumer products like dunks suit homes.[ ppl-ai-file-upload.s3.amazonaws ][ indogulfbioag ] Chemical Practicality Cheaper upfront for large areas, but resistance, regulations, and liabilities increase expenses.[ nitrogenbacteria.wixsite ] Direct Comparison Aspect Bti Chemical Pesticides Cost per Treatment Comparable [ nitrogenbacteria.wixsite ] Often lower initially [ nitrogenbacteria.wixsite ] Long-Term Cost Lower (less resistance) [ nitrogenbacteria.wixsite ] Higher (treadmill) [ pmc.ncbi.nlm.nih ] Application Ease Simple, targeted [ ppl-ai-file-upload.s3.amazonaws ] Broad, drift-prone [ pmc.ncbi.nlm.nih ] Bti wins for sustainability.[ nitrogenbacteria.wixsite ] When to Choose Each Use Bti for routine larval control in sensitive areas, organics, and IVM programs—it's the gold standard per WHO and EPA. Reserve chemicals for emergencies where speed trumps safety, always with monitoring. nature+2 Combining both in rotation maximizes benefits.[ indogulfbioag ] Conclusion and Further Reading Bti outperforms chemicals in safety, ecology, and durability, making it preferable for modern pest management despite slightly slower action. For detailed FAQs on Bacillus thuringiensis israelensis applications and safety, visit: https://www.indogulfbioag.com/microbial-species/bacillus-thuringiensis-israelensisfmicb-14-1293302.pdf+1nature+1
- What is Bacillus Thuringiensis Israelensis used for?
Photo credit: https://www.researchgate.net/figure/B-thuringiensis-subsp-israelensis-spores-in-food-vacuoles-of-T-pyriformis-A-and_fig1_51344908 Bacillus thuringiensis israelensis (Bti) is a biological larvicide used worldwide to control mosquitoes, black flies, and certain other dipteran pests in an environmentally responsible way. It is valued because it targets specific insect larvae without harming humans, pets, wildlife, or beneficial insects when used as directed. indogulfbioag+3 What is Bacillus thuringiensis israelensis? Bacillus thuringiensis subsp. israelensis is a Gram‑positive, spore‑forming soil bacterium first identified in Israel’s Negev Desert in 1977. During sporulation it produces insecticidal crystalline proteins (ICPs) such as Cry4A, Cry4B, Cry11A, and Cyt1A that are toxic to certain fly larvae when ingested. indogulfbioag+2 These crystal proteins dissolve in the alkaline gut of susceptible larvae, bind to receptors in the gut lining, and form pores in the intestinal cells. The damaged gut allows bacteria and gut contents to enter the body cavity, leading to larval death from septicemia or starvation. This highly specific mode of action is why Bti affects only a narrow group of dipteran larvae and is considered safe for non‑target organisms. epa+3 Main uses of Bti 1. Mosquito larval control The primary and best‑known use of Bti is the control of mosquito larvae in water bodies before they emerge as biting adults. Public health agencies, municipalities, and private operators apply Bti to breeding habitats such as ponds, marshes, drainage channels, rice fields, sewage lagoons, storm‑water catch basins, and artificial containers. pmc.ncbi.nlm.nih+3 Target mosquito groups include many species of Aedes, Culex, and Anopheles that transmit diseases like dengue, Zika, chikungunya, West Nile virus, and malaria. By focusing on the larval stage, Bti reduces adult mosquito populations and disease risk without blanket spraying of chemical adulticides over residential areas. In aquaculture and irrigation systems, Bti can be used to suppress mosquito breeding without contaminating fish or crops. rdek+4 2. Control of black flies and other biting midges Bti is also widely used against black fly (Simuliidae) larvae, which develop in flowing water and can cause severe biting nuisance and transmit diseases in some regions. Applications in rivers and streams target larval stages attached to submerged substrates, reducing adult emergence and biting pressure on humans and livestock. indogulfbioag+1 Certain commercial formulations and programs use Bti for other Nematocera such as some midges and fungus gnat larvae, particularly in greenhouse or high‑humidity environments. In these systems, Bti helps protect both workers and plants from nuisance and damage associated with high gnat populations. indogulfbioag+1 3. Larvicide in integrated vector management (IVM) Bti is a cornerstone tool in integrated vector management, where multiple tactics are combined to keep vector populations below harmful levels. It is frequently rotated or combined with other biological agents such as Lysinibacillus (Bacillus) sphaericus to slow resistance development and extend product life. pmc.ncbi.nlm.nih+3 Within IVM, Bti complements environmental management (eliminating standing water), personal protection measures, and, where necessary, targeted chemical control. This layered approach is especially important in regions facing multiple mosquito‑borne diseases and where communities demand safer, more sustainable control solutions. indogulfbioag+2 Agricultural and horticultural uses 4. Use in organic farming and crop environments Because of its specificity and favorable safety profile, Bti is approved for use in organic production systems in many jurisdictions. Organic and conventional growers can use Bti‑based larvicides around irrigation ditches, reservoirs, and crop‑adjacent water bodies to manage mosquito larvae without compromising crop safety or certification status. indogulfbioag+2 Commercial Bti products are also used in protected cultivation and ornamental production to suppress fungus gnat larvae in growing media. These pests can damage roots and transmit plant pathogens; incorporating Bti into integrated pest management programs helps protect root systems while maintaining a low chemical footprint.[ indogulfbioag ] 5. Role in broader biological pest‑control portfolios Bti is often positioned alongside other Bacillus‑based products within biological pest‑control portfolios. While other Bacillus thuringiensis subspecies target caterpillars (Lepidoptera) or beetle larvae (Coleoptera), Bti is the subspecies of choice for dipteran larvae such as mosquitoes and black flies. indogulfbioag+3 Manufacturers integrate Bti into larvicide ranges for public health, animal housing, and environmentally sensitive areas such as wetlands and conservation zones. In this way, Bti helps operators move away from broad‑spectrum synthetic larvicides toward more targeted, residue‑free options. indogulfbioag+4 Environmental and public‑health applications 6. Urban and residential mosquito management Many cities use Bti in neighborhood mosquito‑control programs, treating catch basins, storm drains, roadside ditches, and retention ponds. Granular or briquette formulations can be placed directly into water bodies to release Bti toxins over time, focusing activity where larvae feed. epa+2 Householders and property managers can also use consumer Bti products in birdbaths, rain barrels, ornamental ponds, and other small water features. This helps break the mosquito life cycle close to homes, improving comfort and reducing the need for repeated adulticide spraying. cdc+2 7. Protection of sensitive habitats and wildlife Bti is frequently selected for mosquito control in ecologically sensitive areas such as wetlands, wildlife reserves, and drinking‑water catchments. Decades of research show that, when used according to label directions, Bti has minimal direct impacts on non‑target aquatic invertebrates, fish, birds, mammals, and amphibians. pmc.ncbi.nlm.nih+2 It degrades relatively quickly in the environment, with no long‑term buildup in water or soil, which further limits ecological risk. Some studies investigate possible indirect effects on food webs under very intensive use, so many programs monitor local biodiversity and adjust application strategies accordingly. Overall, though, Bti remains one of the most widely accepted larvicides for conservation areas and drinking‑water sources. opus4.kobv+3 Why Bti is considered safe 8. Human and animal safety Regulators such as the U.S. Environmental Protection Agency classify Bti as posing no known risk to human health when used as directed. Toxicology studies show no evidence of toxicity when Bti is ingested, inhaled, or contacts intact skin at labeled use rates. indogulfbioag+1 Similarly, studies report that Bti is non‑toxic to mammals, birds, fish, and most aquatic invertebrates at operational doses. Occasional mild eye or skin irritation can occur when handling concentrated products, so standard personal protective equipment—gloves, eye protection, and dust masks—is recommended during mixing and application. epa+2 9. Environmental fate and non‑target effects Bti spores and toxins break down within days to weeks in most field conditions, under the influence of sunlight, microbial activity, and dilution. This rapid degradation means Bti does not persist or bioaccumulate in soil and water in the way some synthetic pesticides can. pmc.ncbi.nlm.nih+1 Extensive monitoring and field trials confirm minimal direct effects on pollinators such as bees, beneficial predatory insects, and most non‑target aquatic organisms at labeled rates. Because Bti must be ingested by susceptible larvae and activated in a specific type of alkaline gut, organisms without the right gut conditions and receptors are unaffected. pmc.ncbi.nlm.nih+4 Practical considerations for using Bti 10. Formulations and application methods Bti is formulated as granules, wettable powders, liquid concentrates, and slow‑release briquettes or tablets, each suited to particular habitats and operational needs. Granular and briquette products are common in small containers and catch basins, while liquids and powders are frequently used in large‑scale aerial or ground applications over wetlands and floodplains. rdek+3 For effective control, applicators must match dose to habitat type, water depth, and larval density, and time applications to coincide with early to mid‑larval stages. Label guidance typically specifies avoiding strong winds and temperature inversions to minimize drift and ensure Bti deposits in water where larvae feed. indogulfbioag+3 11. Resistance management and long‑term performance Although Bti uses multiple toxins with different binding sites, resistance is still a theoretical and, in some cases, observed risk when the same agent is used too frequently in isolation. Programs mitigate this by rotating Bti with other microbial larvicides, using combination products, and integrating environmental management to reduce the number of required treatments. pmc.ncbi.nlm.nih+2 Regular monitoring of larval susceptibility and field efficacy helps detect early shifts in sensitivity and supports timely adjustments to control strategies. This proactive resistance management helps preserve Bti as a reliable, long‑term tool in global mosquito‑control campaigns. indogulfbioag+3 Linking to more information on Bti safety For readers who want to explore the safety aspects of Bti in more depth—covering human health, pets, wildlife, and the environment—see the detailed FAQ section on Bti and mosquito control safety provided here:[ indogulfbioag ] https://www.indogulfbioag.com/post/bti-mosquito-control-safety
- Biotechnological Applications of Prodigiosin Produced by Serratia marcescens
In the vibrant red colonies of Serratia marcescens lies one of nature's most versatile bioactive compounds: prodigiosin, a remarkable secondary metabolite that has captivated biotechnologists, pharmaceutical researchers, and industrial scientists for decades. This natural red pigment—with its distinctive chemical formula C₂₀H₂₅N₃O and molecular weight of 323 Da—represents far more than an aesthetic curiosity; it embodies a treasure trove of biotechnological applications ranging from cutting-edge cancer therapeutics to sustainable natural colorants. The biotechnological significance of prodigiosin extends across multiple sectors of contemporary industry and medicine. The pigment exhibits an extraordinary spectrum of biological activities including potent anticancer properties, immunosuppressive effects, broad-spectrum antimicrobial and antifungal capabilities, antioxidant potential, and emerging applications as pH indicators, UV protectors, and biofilm-controlling agents. These multifaceted activities, combined with low toxicity to normal human cells and the capacity for microbial fermentation at industrial scale, position prodigiosin as a promising candidate for addressing critical challenges in oncology, pharmaceutical development, food industry, and environmental remediation. This comprehensive analysis explores the complete spectrum of biotechnological applications of prodigiosin produced by Serratia marcescens , examining the biochemical mechanisms underlying its biological activities, current production methodologies, optimization strategies, purification techniques, and diverse industrial and pharmaceutical applications. The analysis reveals that prodigiosin represents not merely a single-function compound but rather a multidimensional platform technology with expanding applications as research continues to uncover novel biological activities and optimize production processes. Chemical Structure and Fundamental Properties Understanding the biotechnological applications of prodigiosin requires first understanding its chemical structure and inherent properties that enable its diverse biological activities. Chemical Architecture Prodigiosin belongs to the prodiginine family of polypyrrole compounds—a class of alkaloid pigments characterized by a linear tripyrrole chromophore (three pyrrole rings linked in sequence). The specific structure of prodigiosin features: Three interconnected pyrrole rings: Forming the core chromophore responsible for the distinctive red color Methoxy substitution: At position 6 of the prodiginine structure Pentyl side chain: Contributing to hydrophobic properties and membrane interaction Conjugated π-electron system: Across the tripyrrole backbone, providing electronic properties responsible for light absorption (λmax = 534-535 nm) and photochemical reactivity This structural architecture confers fundamental properties: Color Properties: Absorption maximum: 534-535 nm (visible red light) Distinctive red hue enabling visual identification and concentration monitoring Fluorescent properties: Autofluorescence capability providing analytical applications Chemical Solubility: Lipophilic (hydrophobic): Soluble in organic solvents (ethanol, methanol, acetone, toluene) Limited aqueous solubility: Hydrophobic nature constrains direct aqueous application but enables membrane penetration pH-dependent behavior: Structure influenced by solution pH, enabling pH indicator applications Stability Characteristics: Temperature sensitivity: Decomposition increases above 37-40°C, requiring temperature-controlled storage Light sensitivity: UV exposure induces photodegradation, necessitating dark storage Chemical stability: Stability enhanced under acidic conditions Pharmaceutical Applications: Anti-Cancer Potential The most extensively researched and clinically promising biotechnological application of prodigiosin involves cancer therapeutics. Prodigiosin exhibits remarkable anticancer properties through multiple mechanisms while demonstrating minimal toxicity to normal, non-cancerous cells—a critical distinction that separates it from many conventional chemotherapy agents. Anticancer Mechanism of Action Apoptosis Induction Through Multiple Pathways: Prodigiosin triggers programmed cell death (apoptosis) in cancer cells through sophisticated mechanisms: Copper-Mediated DNA Cleavage:The most novel mechanism involves copper ions. Cancer cells accumulate copper at approximately 3.5-fold higher concentrations than non-malignant cells. Prodigiosin chelates copper, forming complexes that cleave DNA at specific sites. This copper-mediated mechanism explains prodigiosin's selective toxicity to cancer cells—cancer cells contain sufficient copper to activate the mechanism, while normal cells do not. The DNA cleavage mechanism involves: Prodigiosin-copper complex formation Oxidative damage to DNA strands Activation of apoptotic cascades Cancer cell death without normal cell toxicity p53 Pathway Modulation:Many cancer cells harbor mutations in the p53 tumor suppressor gene, eliminating critical apoptotic mechanisms. Prodigiosin rescues defective p53 pathways by: Upregulating p73 (a p53 homolog) Targeting mutant p53/p73 interactions Restoring apoptotic capacity in p53-mutant cancer cells This mechanism is particularly valuable for treating multidrug-resistant (MDR) cancers with p53 defects, which typically show poor chemotherapy response. mTOR Pathway Inhibition:Prodigiosin inhibits the mammalian target of rapamycin (mTOR) pathway—a critical pathway controlling cell growth and proliferation. mTOR inhibition: Suppresses protein synthesis Induces cell cycle arrest Triggers autophagy in cancer cells Prevents angiogenesis (blood vessel formation supplying tumors) HSP90 Downregulation:Heat shock protein 90 (HSP90) is essential for cancer cell survival, particularly in triple-negative breast cancer (TNBC). Prodigiosin downregulates HSP90α expression, undermining cancer cell survival mechanisms and sensitizing resistant tumors to other therapies. Clinical Evidence: Documented Efficacy Cancer Cell Line Studies: Laboratory studies demonstrate remarkable efficacy against diverse cancer types: Cancer Type Cell Line Prodigiosin Efficacy Notes Breast Cancer MDA-MB-231 Cytotoxic at low concentrations Triple-negative cancer; notoriously resistant Breast Cancer MCF-7 Strong inhibitory ability Estrogen receptor-positive cancer Colon Cancer HT-29 IC50: 47 μM Potent cell growth suppression Lung Cancer LU-1 Strong inhibitory ability Notoriously difficult to treat Melanoma SK-MEL-30 IC50: 70 μM Multidrug-resistant skin cancer Neuroblastoma SH-SY5Y Selective toxicity Protective antioxidant effects in normal neurons Leukemia Human T-cell leukemia Apoptosis induction Acute leukemia response documented In Vivo Tumor Studies: Animal model studies demonstrate clinical relevance: Serratia marcescens QBN VTCC 910026 prodigiosin in tumor-bearing mice: Tumor volume reduction: 36.82% after 28 days of treatment Survival improvement: Extended survival compared to untreated controls Efficacy comparable to conventional chemotherapy: Achieved through biological rather than chemical mechanisms These results indicate clinical potential for advanced development. Prodigiosin Derivatives in Clinical Trials: Pharmaceutical companies have advanced prodigiosin-based compounds into clinical development: Obatoclax (GX15-070): Prodigiosin derivative showing potent anti-cancer and pro-apoptotic properties Clinical trials: Efficacy demonstrated in treating: Relapsed chronic lymphocytic leukemia (CLL) Extensive-stage small cell lung cancer Therapeutic outcomes: Positive results both as monotherapy and in combination regimens Safety profile: Good tolerability without documented genotoxicity The advancement of prodigiosin analogs into clinical trials represents validation of the compound's therapeutic potential. Selectivity for Cancer Cells: Why Normal Cells Are Spared Critical to prodigiosin's pharmaceutical value is its selective toxicity to cancer cells—cytotoxic activity against malignant cells while sparing normal, healthy cells. This selectivity derives from fundamental differences between cancer and normal cell biology: Cancer Cell Characteristics That Enable Prodigiosin Toxicity: Elevated intracellular copper accumulation Defective p53 pathway requiring rescue mechanisms Hyperactive mTOR signaling Elevated HSP90 dependency High metabolic rates supporting copper homeostasis requiring prodigiosin interaction Normal Cell Protection Mechanisms: Lower intracellular copper concentrations (insufficient for prodigiosin-copper DNA cleavage) Intact p53 pathways rendering p73 upregulation unnecessary Balanced mTOR signaling (not cancer-dependent) Lower HSP90 dependency Metabolic profiles that reduce prodigiosin susceptibility This selective toxicity—demonstrated across numerous cancer cell lines—positions prodigiosin as a biologically-rational anticancer strategy superior to chemotherapies that indiscriminately damage both malignant and normal cells. Immunomodulatory Applications Beyond direct anticancer activity, prodigiosin functions as a sophisticated immunomodulator—reshaping immune system responses in ways that support cancer treatment and transplant tolerance. Mechanism of Immunomodulation Immune Cell Reprogramming: Prodigiosin modulates immune cells within the tumor microenvironment (TME)—the complex cellular ecosystem surrounding cancer: T Cell Modulation: Induces apoptosis in T and B lymphocytes Alters T cell differentiation pathways Modulates IL-10 and IL-4 serum levels (anti-inflammatory cytokines) Reduces immunogenic T cell responses that previously failed to control cancer Macrophage Reprogramming: Influences tumor-associated macrophages (TAMs) Shifts macrophage phenotype from cancer-supporting to cancer-inhibiting Reduces immunosuppressive factor production NK Cell and Dendritic Cell Effects: Modulates natural killer (NK) cell function Influences tumor-associated dendritic cells (TADCs) Reduces myeloid-derived suppressor cell (MDSC) populations Programmed Death Ligand (PD-L1) Modulation:Prodigiosin downregulates PD-L1—a checkpoint protein through which cancer cells evade immune surveillance. By reducing PD-L1, prodigiosin restores anti-tumor immune responses, potentially enhancing efficacy of immune checkpoint inhibitors. Clinical Applications in Immunotherapy The immunomodulatory properties create multiple clinical applications: Combination with Checkpoint Inhibitors: Prodigiosin + anti-PD-1/PD-L1 antibodies: Synergistic immune activation Enhanced anti-tumor immunity through complementary mechanisms Potential to overcome resistance to checkpoint inhibitors Transplantation Tolerance: Enhanced survival in mice undergoing heart transplantation Immunosuppressive effects preventing rejection Potential application in solid organ transplantation Alternative to conventional immunosuppressants with different toxicity profiles Synergistic Therapy: Prodigiosin shows additive effects with rapamycin Synergistic effects with cyclosporin A Potential for combination regimens optimizing immune modulation Antimicrobial and Antifungal Applications Beyond cancer and immunotherapy, prodigiosin exhibits broad-spectrum antimicrobial and antifungal activities with applications in medical treatment, food preservation, and environmental remediation. Antimicrobial Efficacy Bacterial Sensitivity: Prodigiosin demonstrates bactericidal and bacteriostatic activity against diverse pathogenic bacteria: Bacterial Species Minimum Inhibitory Concentration (μg/mL) Minimum Bactericidal Concentration (μg/mL) Clinical Significance Oxacillin-resistant E. coli 1 2 Multidrug-resistant infection Pseudomonas aeruginosa 2 4 Hospital-acquired infections Oxacillin-resistant S. aureus (ORSA) 4 8-16 MRSA infections Staphylococcus aureus <4 Variable Skin/soft tissue infections Streptococcus pyogenes <4 Variable Group A Strep infections Enterococcus faecalis <4 Variable Antibiotic-resistant UTIs Acinetobacter sp. <4 16 Nosocomial infections These minimum inhibitory concentrations demonstrate potent antimicrobial activity at remarkably low concentrations, comparable to or exceeding conventional antibiotics. Mechanism of Antimicrobial Action: Prodigiosin disrupts bacterial cell membranes through: Lipid bilayer destabilization Ion leakage (particularly potassium) Membrane depolarization Osmotic imbalance leading to cell death The multi-target mechanism makes resistance development extremely unlikely—bacteria cannot survive without functional cell membranes. Antifungal and Oomyceticide Activity Prodigiosin demonstrates remarkable activity against fungal pathogens: Fungal Pathogen Sensitivity: Candida species: Susceptible to prodigiosin Aspergillus species: Antifungal activity documented Rhizoctonia solani : Agricultural fungal pathogen suppressed Phytophthora infestans : Oomycete pathogen (late blight) inhibited Fusarium species: Plant pathogenic fungus suppressed Specific Antifungal Mechanisms: Chitin synthesis inhibition Ergosterol binding disrupting membrane structure Oxidative damage to fungal cells Interference with fungal protein synthesis Food Preservation Applications The antimicrobial and antioxidant properties make prodigiosin valuable in food applications: Preservative Function: Prodigiosin demonstrates dual-purpose preservation: Antimicrobial preservation: Inhibits foodborne pathogens Antioxidant preservation: Prevents oxidative rancidity through free radical scavenging Field trials demonstrate: Prodigiosin-treated food products: Extended shelf life vs. untreated controls Foodborne pathogen suppression: Effective against spoilage organisms Free radical scavenging: 99% inhibition of DPPH radicals at 10 μg/mL concentration ABTS radical scavenging: >95% activity Antioxidant Mechanism: Prodigiosin scavenges free radicals through: Hydrogen atom donation to alkyl radicals Chelation of pro-oxidant metal ions Regeneration of α-tocopherol and ascorbate Electron transfer to reactive oxygen species The free radical scavenging activity positions prodigiosin as a natural antioxidant preservative superior to synthetic alternatives. Current Limitations in Food Industry: Despite antimicrobial and antioxidant potential, regulatory approval for direct food additives remains limited pending comprehensive toxicological assessment. However, authorization progression is likely given favorable safety profiles documented to date. Dye and Colorant Applications Prodigiosin's distinctive red hue and vibrant color properties make it valuable as a natural colorant for diverse industries—from textiles to cosmetics to food applications. Textile Dyeing Dyeability Across Fabric Types: Industrial trials demonstrate prodigiosin's textile application potential: Fabric Type Color Quality Shade Intensity Conditions Bubble cotton Excellent Deep red Acidic, basic, neutral Lycra Excellent Deep red All pH conditions Cotton 20/20 blend Excellent Deep red All conditions Hosiery Excellent Deep red All conditions Cotton polyester Good Moderate pH-dependent Cotton Good Moderate Moderate intensity Polyester Moderate Light Limited affinity Silk Moderate Light Light shades Linen Poor Faint Minimal staining Advantages Over Synthetic Dyes: Natural source (microbiologically produced) Biodegradable (unlike persistent synthetic colorants) Absence of carcinogenic byproducts (many synthetic dyes removed due to carcinogenic azo compounds) Multiple biological activities (antimicrobial properties in dyed fabrics) pH Behavior:Prodigiosin demonstrates pH-dependent color shifts enabling applications as a pH indicator dye—with color changes corresponding to solution acidity/alkalinity. This property enables applications in: pH indicator paper Acid-base indicator systems Real-time pH monitoring in industrial processes Cosmetic and Personal Care Applications The cosmetic industry represents a major market opportunity for prodigiosin: Cosmetic Pigment Market: Global market size (2022): ~$700 million Projected growth (2032): $1.5+ trillion Prodigiosin positioned for significant penetration given biological activity advantage Cosmetic Applications: Nail Polish Colorant: Provides vibrant red shade Biological antimicrobial activity inhibits fungal nail infections Natural alternative to synthetic colorants Lipstick and Lip Tints: Natural red pigmentation Antimicrobial properties preserve product Antioxidant activity protects lip tissue Face Cosmetics: Foundations, blushes, eyeshadows UV protection properties (emerging application) Antioxidant activity protecting skin from oxidative damage Skincare Products: Creams and serums with prodigiosin-enhanced formulations Antioxidant benefits supporting skin health Antimicrobial properties supporting skin barrier function Regulatory Status:Cosmetic applications face fewer regulatory barriers than pharmaceutical or food applications, enabling faster market entry with established safety profiles. Food Industry Applications Natural Food Colorant: Prodigiosin demonstrates potential as a food-safe natural colorant: Current Applications: Bakery product decoration: Cakes, pastries, confections Candy coloring: Natural alternative to synthetic dyes Beverage coloring: Soft drinks, juices, alcoholic beverages Ice cream coloring: Natural food-grade pigmentation Advantages of Prodigiosin as Food Colorant: Natural origin (not synthetic chemical) Antimicrobial co-benefit (extends product shelf life) Antioxidant activity (prevents oxidative deterioration) No documented adverse health effects at food-relevant concentrations Biodegradable (unlike persistent synthetic colorants) Regulatory Pathway:Comprehensive toxicological testing underway to establish safety for direct food additives. Likely approval trajectory given favorable safety data positions prodigiosin for significant food industry penetration. Paper, Inks, and Specialty Applications Paper Coloration: Paper dyeing for specialty applications Prodigiosin-dyed paper serves as natural pH indicator paper Artistic applications requiring natural pigmentation Industrial Ink: Highlighter pen ink formulations: >0.25% prodigiosin concentration shows promising results Biodegradable ink alternative to synthetic formulations Specialized labeling inks with antimicrobial properties Soap and Candle Manufacturing: Natural colorant for soap products (1-3 mg/L concentration) Candle coloration with variable intensities Natural fragrance/color combination in artisanal applications Emerging Applications: pH Indicators, UV Protection, and Biosensors Beyond established pharmaceutical and industrial uses, emerging applications continue to expand prodigiosin's utility: pH Indicator Applications The pH-dependent color changes of prodigiosin enable novel applications: pH Indicator Paper: Prodigiosin-dyed paper shows color changes corresponding to pH Natural alternative to synthetic pH indicators Applications in educational, laboratory, and field testing Real-Time pH Monitoring: Integration in smart packaging Indicators for food freshness/pH monitoring Quality control in industrial processes Biological pH Sensing: Fluorescence-based pH sensing Integration in biosensors Medical diagnostic applications UV Protection Applications Emerging research reveals prodigiosin's capacity as a UV protective agent: Mechanism: Absorption of UV radiation preventing skin damage Antioxidant activity protecting against UV-induced free radical damage Integration in sunscreen formulations Cosmetic protective effect against solar radiation Cosmetic Applications: Sunscreen and UV protection formulations Daily skincare products with UV protection Natural alternative to synthetic UV absorbers Antioxidant Supplement Applications The documented free radical scavenging activity (up to 99% at appropriate concentrations) positions prodigiosin for health supplement applications: Health Claims Under Investigation: Antioxidant supplement supporting cellular health Potential applications in aging-related conditions Cardiovascular health support through antioxidant mechanisms Neuroprotection in neurodegenerative disease models Regulatory Status:Nutraceutical and supplement applications require safety and efficacy documentation. Ongoing research establishing dosage requirements and long-term safety will determine commercial viability. Production and Optimization: Industrial Biotechnology The commercial viability of prodigiosin applications depends on economically feasible, large-scale production. Industrial biotechnology continues advancing production methodologies and optimization strategies. Fermentation Optimization Optimal Growth Conditions: Research has identified conditions maximizing prodigiosin production: Temperature Optimization: Optimal temperature: 22-30°C (varies by strain) Temperature sensitivity: Production decreases above 37°C Strain-specific optimization: Gamma-irradiated strains functional up to 36°C pH Optimization: Optimal pH: 9.0 (neutral pH reduces production) pH sensitivity: Production regulated by multiple sensor systems Nutrient Optimization: Peptone concentration: 1% (w/v) optimal Inoculum size: 10⁹ cells/mL Incubation period: 6-7 days for maximum production (72 mg/L typical) Medium Composition: Innovative media formulations utilizing agricultural wastes: Wheat bran: Solid-state fermentation achieving 998.3 mg/kg/h productivity Tannery fleshing: Combined with wheat bran, achieving 2,963.7 mg/kg/h productivity Crude glycerol (from biodiesel): Economical carbon source for fermentation Rice straw: Sustainable feedstock enabling 2,440 mg/L production Strain Development and Genetic Engineering Hyper-Producing Strains: Conventional mutagenesis and genetic engineering enhance production: Gamma Radiation Mutagenesis: Gamma irradiation (200 Gy dose): Doubles prodigiosin production Hyper-producing strains: Functional at elevated temperatures (up to 36°C) Mutation creates favorable phenotype without stability problems Genetic Engineering Approaches: Metabolic engineering enhances yield: Transcription Factor Engineering: Overexpression of OmpR and PsrA regulators: Increases prodigiosin synthesis genes expression Recombinant strain PG-6: Enhanced production (10.25 g/L vs. 6.33 g/L wild-type) 1.62-fold improvement in prodigiosin titer achieved Quorum Sensing Pathway Engineering: Manipulation of AI-1 (acyl-homoserine lactone) autoinducer signaling Optimization of LuxS/AI-2 metabolic pathways Enhanced regulation of pig gene cluster expression Promoter Engineering: Identification of strong, constitutive promoters (P17 promoter) Replacement of native pig gene promoters with stronger alternatives Increased metabolic flux through prodigiosin biosynthesis pathway Industrial Scale Production and Bioreactors Bioreactor Scale-Up: Production strategies for commercial scale: Batch Fermentation: Standard 10-100 L bioreactors Maximum PG productivity: 998.3 mg/kg/h substrate (wheat bran) Large-Scale Production: 200 L bioreactors: Industrial-scale demonstration Typical yields: 60-72 mg/L in optimized conditions Production cycle: 72-120 hours fermentation Advanced Bioreactor Designs: Extraction columns with HP-20 adsorbent resin: 31% increase vs. batch culture Improved oxygen transfer rate (OTR) optimization Fed-batch strategies enabling higher cell densities Economic Considerations: Cost optimization strategies: Waste utilization: Crude glycerol, wheat bran, tannery wastes reduce feedstock costs Strain optimization: Engineered strains increase yield per unit feedstock Downstream simplification: Improved extraction/purification reduces processing costs Current production cost estimates: Competitively positioned with synthetic colorants for market entry Extraction, Purification, and Analytical Characterization Commercial application requires efficient, scalable purification protocols producing pharmaceutical-grade prodigiosin. Extraction Methodologies Solvent-Based Extraction: Extraction from bacterial culture: Direct Extraction from Culture: Acetone extraction: Dissolves prodigiosin from bacterial cells Methanol extraction: Alternative solvent with good extraction efficiency Ethanol extraction: Pharmaceutical-grade extraction solvent Recovery from Media: Centrifugation: Separation of cell pellet and supernatant (15,000 rpm, 20 min) Both cell-associated and extracellular prodigiosin recovered separately Combined extraction from both fractions maximizes yield Solvent Selection for Purification: Selective solvent systems enable purification: Toluene:ethyl acetate (9:1 v/v): High selectivity for prodigiosin Chloroform:ethyl acetate (1:1 v/v): Alternative system for purity Petroleum ether:ethyl acetate gradients: Silica gel chromatography mobile phases Purification Techniques Column Chromatography: Multi-stage purification achieving high purity: Silica Gel Column Chromatography: Initial separation using silica gel resin Gradient elution with petroleum ether:ethyl acetate ratios Collection of red fractions at specific solvent ratios Preliminary purification achieving 70-80% purity Glass Column Chromatography (HP-20 Resin): Secondary purification step Removal of hydrophilic contaminants Washing with aqueous solutions removes salts/sugars Elution with ethanol concentrates prodigiosin Preparative HPLC: High-performance liquid chromatography for final purification: Analytical HPLC Parameters: Detection wavelength: 535 nm (maximum absorbance) Reference wavelength: 630 nm (baseline correction) Alternative detection: 254 nm Mobile phases: 0.1% formic acid in water (solvent A) and methanol (solvent B) Isocratic or gradient elution depending on purity requirements Preparative HPLC: Large-scale purification columns Collection of prodigiosin-containing fractions Final purity achievement: >98% (HPLC-confirmed) Typical recovery: ~360 mg purified prodigiosin from 35 g crude extract Analytical Characterization and Quality Assurance Chemical Identity Confirmation: Multiple analytical techniques confirm prodigiosin identity: UV-Visible Spectroscopy: λmax = 534-535 nm (distinctive absorption) Unique color providing visual confirmation Quantification via Beer-Lambert law High-Performance Liquid Chromatography (HPLC): Retention time comparison with prodigiosin standard Purity determination (>95% threshold for pharmaceutical use) Quantitative measurement of prodigiosin concentration Mass Spectrometry (MS): Molecular ion peak: m/z = 323 (matching molecular weight C₂₀H₂₅N₃O) Diagnostic fragments confirming structure LC-MS analysis of purified extracts Nuclear Magnetic Resonance (NMR): ¹H-NMR spectroscopy: Confirms tripyrrole structure ¹³C-NMR spectroscopy: Validates carbon skeleton Chemical shift patterns characteristic of prodigiosin Fourier Transform Infrared (FT-IR) Spectroscopy: Identification of characteristic functional groups Pyrrole C-H stretching patterns Comparative analysis with reference standards Chromatographic Purity: Thin-layer chromatography (TLC): Rapid purity assessment Gas chromatography-mass spectrometry (GC-MS): Organic contaminant detection Ultra-performance liquid chromatography (UPLC): High-resolution purity analysis Limitations and Challenges: Honest Assessment Despite remarkable potential, prodigiosin development faces significant challenges requiring continued research and optimization: Hydrophobicity and Bioavailability Challenge:Prodigiosin's lipophilic nature, while enabling membrane penetration and tissue interaction, creates pharmaceutical challenges: Aqueous solubility limitation: Poor aqueous solubility constrains direct administration Formulation complexity: Requires pharmaceutical excipients and nanoformulation strategies Bioavailability variability: Tissue distribution dependent on formulation approach Solutions Under Development: Nanoparticle formulations (liposomes, nanospheres) Bionanocomposites improving bioavailability Prodrug development enhancing solubility Combination with bioavailability enhancers Stability and Storage Requirements Challenge:Temperature and light sensitivity constrains product shelf-life and distribution: Temperature sensitivity: Decomposition above 37°C Photodegradation: UV exposure induces structural breakdown Stability limited: Requires dark, temperature-controlled storage Solutions: Protective formulations (encapsulation, stabilizing excipients) Storage in inert atmosphere packages Refrigerated distribution systems Antioxidant co-formulation preventing oxidative degradation Production Costs and Economic Viability Challenge:Current production costs compete with synthetic alternatives: Fermentation costs: Labor-intensive, temperature-controlled bioprocesses Downstream processing: Extraction and purification represent significant expense Market competition: Synthetic dyes and pharmaceuticals offer price competition Improvements Enabling Competitiveness: Waste feedstock utilization reducing raw material costs Strain optimization increasing productivity per unit feedstock Automated bioprocessing reducing labor costs Regulatory approval enabling premium pricing for pharmaceutical applications Regulatory and Safety Considerations Challenge:Food and pharmaceutical applications require extensive safety documentation: Toxicological testing: Long-term safety studies required before regulatory approval Genotoxicity assessment: Potential genetic damage must be excluded Pharmacokinetics: Absorption, distribution, metabolism, excretion characterization needed Clinical trials: Pharmaceutical applications require full clinical development pathways Current Status: Favorable genotoxicity profiles documented in preliminary studies Prodigiosin analogs advanced into clinical trials demonstrate safety Expansion of approved applications dependent on additional safety data Timeline to widespread pharmaceutical approval: 5-10+ years depending on indication Market Opportunities and Commercial Trajectory Despite current limitations, biotechnological applications of prodigiosin represent substantial commercial opportunities: Pharmaceutical Market Cancer Therapeutics Market: Global oncology pharmaceutical market: >$200 billion annually Natural product-derived drugs: Growing segment (25% of new FDA approvals) Prodigiosin-based therapeutics: Potential multi-billion dollar market opportunity Timeline to market: 2028-2035 for advanced candidates Industrial Pigment Market Natural Colorant Market: Global natural colorant market: ~$1.5 billion (2023) Growth rate: 7-10% CAGR Prodigiosin positioning: Premium-priced natural colorant with biological co-benefits Market segments: Textiles, cosmetics, food industry, specialty inks Food Industry Applications Natural Food Additive Market: Global food additives: >$60 billion annually Natural/organic segment: Fastest-growing (15%+ CAGR) Dual-function preservative + colorant: Valuable market position Timeline to market: 2026-2030 pending regulatory approval Conclusion: Prodigiosin as a Multidimensional Biotechnology Platform Prodigiosin produced by Serratia marcescens represents far more than a simple red pigment; it embodies a multidimensional biotechnology platform with applications spanning oncology, immunotherapy, antimicrobiology, food science, cosmetics, industrial chemistry, and environmental remediation. The compound's remarkable spectrum of biological activities—potent anticancer effects with selective toxicity to malignant cells, immunomodulatory properties enhancing cancer immunotherapy, broad-spectrum antimicrobial and antifungal capabilities, powerful antioxidant activity, and emerging applications as pH indicators and UV protectors—collectively position prodigiosin as a valuable asset for addressing critical challenges in human health and industry. Recent advances in: Genetic engineering (achieving 10+ g/L production in engineered strains) Fermentation optimization (utilizing waste feedstocks reducing costs) Purification technology (achieving >98% purity at industrial scale) Regulatory pathways (prodigiosin analogs in clinical trials) ...create realistic timelines for pharmaceutical approval and commercial market penetration. While challenges remain—particularly aqueous solubility, thermal stability, production economics, and regulatory approval—these represent surmountable barriers rather than fundamental limitations. The convergence of scientific evidence (documenting safety and efficacy), technological advancement (enabling cost-effective production), and market opportunity (growing demand for natural bioactive compounds) suggests that prodigiosin-based therapeutics and industrial applications will become increasingly prevalent in pharmaceutical and industrial sectors over the next decade. Serratia marcescens prodigiosin exemplifies how microbial secondary metabolism, harnessed through biotechnology, can provide solutions to challenging problems in human health and industry—demonstrating the continuing relevance of microorganisms as sources of bioactive compounds with profound applications in contemporary medicine and industry. Frequently Asked Questions What are the biotechnological applications of the prodigiosin pigment produced by Serratia marcescens? Prodigiosin, the vibrant red pigment produced by Serratia marcescens , exhibits a remarkably diverse spectrum of biotechnological applications. The compound has attracted significant interest in biotechnology due to its diverse biological activities, which include: Pharmaceutical Applications: Anticancer properties: Triggers apoptosis in cancer cells through copper-mediated DNA cleavage, p53 pathway modulation, mTOR inhibition, and HSP90 downregulation, with demonstrated efficacy in breast cancer, colon cancer, lung cancer, melanoma, and other malignancies. Prodigiosin derivatives (e.g., Obatoclax) have advanced into clinical trials for chronic lymphocytic leukemia and small cell lung cancer, showing positive therapeutic outcomes. Immunosuppressive effects: Modulates immune responses useful in transplantation tolerance and cancer immunotherapy applications Immunomodulatory activities: Reprograms immune cells in tumor microenvironments, enhancing anti-tumor immunity when combined with checkpoint inhibitors Antimicrobial and Antifungal Applications: Broad-spectrum antimicrobial activity: Exhibits bactericidal effects against methicillin-resistant Staphylococcus aureus , Pseudomonas aeruginosa , Acinetobacter species, and other pathogenic bacteria Antifungal capabilities: Suppresses Candida , Aspergillus , Fusarium , and other fungal pathogens, with particular efficacy against Phytophthora infestans (late blight) Food preservation: Dual-function preservative combining antimicrobial activity with antioxidant effects, extending shelf-life while inhibiting foodborne pathogens Industrial and Cosmetic Applications: Natural dye and colorant: Produces vibrant red color suitable for textiles, cosmetics, bakery products, candles, paper, and specialty inks. Provides natural alternative to synthetic colorants, particularly azo dyes with documented carcinogenic properties. Antioxidant agent: Scavenges free radicals (up to 99% DPPH radical inhibition), supporting applications in food preservation, cosmetic formulations, and health supplements pH indicator: pH-dependent color changes enable applications in pH indicator paper, smart packaging, and real-time monitoring systems UV protector: Emerging application in sunscreen formulations and cosmetic protection Antimicrobial cosmetic additive: Integration in skincare, nail polish, and personal care formulations providing biological co-benefits Research is ongoing to optimize its production through fermentation optimization, genetic engineering of Serratia marcescens strains, and the development of efficient extraction and purification techniques. The goal is to make its production economically viable for diverse pharmaceutical, industrial, and food industry applications. Learn more about Serratia marcescens applications by exploring the detailed product information page , where you'll discover comprehensive guidance on production methodologies, strain optimization, application development, and commercial opportunities for this versatile biotechnological platform organism.
- Can Serratia marcescens Be Used as a Biocontrol Agent in Agriculture? A Comprehensive Guide to Biological Disease Management
By de:Benutzer:Brudersohn - German Wikipedia: http://de.wikipedia.org/wiki/Bild:SerrmarcKol.jpg, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=732821 Introduction In the face of mounting pesticide resistance, environmental contamination, and regulatory restrictions on synthetic fungicides, global agriculture urgently seeks sustainable alternatives to chemical disease management. Among the most promising biological solutions emerging from contemporary agricultural microbiology is Serratia marcescens , a naturally occurring bacterium with remarkable biocontrol capabilities spanning fungal pathogens, plant-parasitic nematodes, and insect pests. The answer to the central question is unambiguous: Yes, Serratia marcescens can be effectively used as a biocontrol agent in agriculture, with field-demonstrated efficacy comparable to or exceeding many synthetic fungicides while offering substantial environmental, health, and sustainability advantages. This bacterial biocontrol agent operates through multiple sophisticated mechanisms—enzymatic degradation of pathogen cell walls, production of broad-spectrum antimicrobial compounds, induction of plant systemic resistance, and biofilm-mediated protection—that collectively create comprehensive disease suppression across diverse crop systems and pathogenic agents. This comprehensive analysis examines the scientific evidence supporting Serratia marcescens as a biocontrol agent, the specific pathogens it controls, the precise mechanisms underlying its effectiveness, practical application methodologies, integration with existing agricultural practices, and the realistic expectations for its role in sustainable disease management. The evidence demonstrates that Serratia marcescens represents not merely another biocontrol option, but rather a multifunctional biological agent capable of addressing multiple agricultural challenges simultaneously—disease suppression, plant growth promotion, stress tolerance enhancement, and nutrient cycling improvement. What is Serratia marcescens? Biological Profile and Agricultural Significance Serratia marcescens is a gram-negative, aerobic bacterium ubiquitous in soil, water, and plant environments worldwide. The organism's name derives from its production of a distinctive prodigiosin pigment—a vibrant red compound that both defines its identity and provides clues to its remarkable biological properties. Key Characteristics Morphological and Taxonomic Features: Rod-shaped (0.8-1.0 μm × 1.5-3.0 μm) gram-negative bacterium Motile (peritrichous flagella enable movement through soil) Facultative anaerobe (metabolically flexible regarding oxygen availability) Non-pathogenic to plants (unlike some bacterial pathogens) Ubiquitous environmental distribution (isolated from 90% of soil samples globally) Pigment and Antimicrobial Production: Prodigiosin: Distinctive red pigment with antimicrobial, anti-inflammatory, and immunosuppressive properties Serrawetin W1: Antimicrobial and antitumor lipopeptide compound Pyrrolnitrin: Broad-spectrum antibiotic with antifungal activity Chitinase and proteases: Enzymatic systems degrading pathogen cell walls Metabolic Versatility: Nitrogen-fixing capacity (some strains) Phosphate solubilization capability (enhancing nutrient availability) Biofilm formation (creating protective communities on plant surfaces) Siderophore production (competing with pathogens for iron) Agricultural Significance and Advantages The agricultural significance of Serratia marcescens derives from the convergence of multiple beneficial properties: Non-pathogenic status: Unlike many bacteria that cause disease under certain conditions, S. marcescens strains used in agriculture are reliably non-pathogenic to plants and safe for human consumption Naturally occurring: Environmental isolation from diverse ecosystems demonstrates established ecological integration Multi-mechanism activity: Simultaneous pathogen suppression, plant growth promotion, and stress tolerance enhancement Broad-spectrum efficacy: Controls fungi, oomycetes, and nematodes—addressing multiple disease vectors simultaneously Pathogenic Targets: Spectrum of Controlled Diseases Scientific studies document Serratia marcescens biocontrol efficacy against an impressive array of agriculturally significant pathogens, spanning multiple pathogen groups and crop systems. Fungal Pathogen Control Serratia marcescens effectively suppresses numerous fungal diseases affecting globally important crops: Soil-Borne Fungal Pathogens: Rhizoctonia solani (Rhizoctonia Rot, Damping-Off) Causative agent of major seed and seedling disease Affects approximately 200 plant species Economic impact: Billions annually in crop losses S. marcescens efficacy: 65-75% disease suppression documented in field trials Fusarium spp. (Fusarium Wilt, Root Rot, Seedling Blight) One of agriculture's most destructive fungal genera Affects tomato, cucumber, banana, wheat, and countless other crops Resistance to chemical fungicides increasingly common S. marcescens efficacy: 60-70% disease suppression, particularly effective in preventive applications Pythium ultimum (Pythium Damping-Off) Aquatic fungus causing seedling disease in diverse crops Particularly damaging in hydroponic and greenhouse systems S. marcescens efficacy: 65-72% suppression, with residual effects throughout growing season Seed Coat and Seedling Diseases:Cucurbits (melon, cucumber, squash) suffer severe losses to Pythium ultimum and Rhizoctonia solani . S. marcescens seed treatments provide: Seed germination protection (67-75% efficacy) Seedling disease suppression (60-70% control) Long-term root colonization providing residual disease suppression Foliar and Aerial Pathogens: Phytophthora infestans (Late Blight, Potato and Tomato) One of agriculture's most economically destructive pathogens Historical significance: Irish potato famine causative organism Chemical resistance increasing Recent research (2025): S. marcescens YNAU-SM-1 strain demonstrates remarkable efficacy: Preventive treatment: 67.62% disease control Simultaneous treatment: 65.48% disease control Curative treatment: 71.04% disease control Sporangial direct germination inhibition: 98.86% Zoospore release inhibition: 70.13% Mechanism Insight: The bacterium produces metabolites that directly inhibit oomycete spore germination—a novel mechanism not exhibited by most chemical fungicides. Plant-Parasitic Nematode Control Serratia marcescens demonstrates potent activity against economically devastating plant-parasitic nematodes affecting global agriculture: Root-Knot Nematodes ( Meloidogyne spp.) Most economically destructive plant-parasitic nematodes globally Cause estimated $157 billion annual agricultural losses Affect >5,000 plant species across all climates Chemical nematicide options increasingly restricted S. marcescens mechanisms: Protease production: Degrades nematode cuticles Direct parasitism: Colonizes nematode body cavities Toxin production: Secondary metabolites inhibit nematode mobility and feeding Rhizosphere competition: Depletes resources nematodes require Plant resistance induction: Enhances host plant defenses Field efficacy data: Root-knot nematode population reduction: 40-60% compared to untreated Plant growth improvement: 25-45% yield increase despite nematode presence Residual effects: Protection maintained throughout growing season Cyst Nematodes ( Heterodera spp. and Globodera spp.) Major pathogens of potato, wheat, soybean Chemical control options limited S. marcescens population suppression: 35-55% reduction Combined with other biocontrol agents: 60-75% suppression Migratory and Semi-Endoparasitic Nematodes: Radopholus similis (burrowing nematode): Banana, plantain pathogen S. marcescens nematicidal activity documented Combination with fungal biocontrol agents (e.g., Pochonia chlamydosporia ): Synergistic suppression Insect Pest Control Emerging research demonstrates that Serratia marcescens functions as an entomopathogenic bacterium—capable of parasitizing and killing insect pests while simultaneously promoting plant growth: Rice Brown Planthopper ( Nilaparvata lugens ) Major rice pest affecting Asia-Pacific region Economic losses: Hundreds of millions annually Endophytic colonization study (2022): S. marcescens S-JS1 seed inoculation demonstrates: Seed germination: +9.4-13.3% Root length: +8.2-36.4% Shoot length: +4.1-22.3% Root fresh weight: +26.7-69.3% Shoot fresh weight: +19.0-49.0% Enhanced secondary metabolite production (conferring pest resistance) Mechanism: Dual-mode action where the bacterium: Directly infects and kills insect pests through entomopathogenic activity Colonizes plant tissues (endophytically) promoting plant vigor and inducing defense gene expression Other Insect Pests: Aphids: Documented suppression through metabolite production Whiteflies: Emerging evidence of biocontrol potential Thrips: Antimicrobial compounds creating hostile feeding environment Mechanisms of Biocontrol: How Serratia marcescens Controls Pathogens The effectiveness of Serratia marcescens as a biocontrol agent derives from multiple sophisticated mechanisms operating simultaneously, creating redundant suppression pathways that pathogens struggle to overcome. Mechanism 1: Enzymatic Degradation and Direct Antagonism Chitinase Production and Activity: Serratia marcescens is among the highest chitinase-producing microorganisms identified. The bacterium secretes chitinase enzymes—molecular machines that catalyze the hydrolysis of chitin, a fundamental component of fungal cell walls and nematode cuticles. Structural Target: Chitin comprises 20-40% of fungal cell wall dry weight and forms the primary structural component of nematode cuticles. Chitinase cleavage of chitin polymers progressively weakens structural integrity: Initial attack: Creates porosity in cell walls Progressive degradation: Widens pores until rupture occurs Cell wall collapse: Pathogen death through osmotic imbalance Enzyme Kinetics: Purified S. marcescens chitinase demonstrates activity across diverse pathogenic fungi: Rhizoctonia solani : 75-85% growth inhibition Bipolaris sp.: 70-80% growth inhibition Alternaria raphani , Alternaria brassicicola : 65-75% growth inhibition Synergistic Enzymatic Systems: Beyond chitinase, S. marcescens produces complementary enzymatic systems: Proteases: Degrade pathogenic proteins and structural components Cellulases: Attack cellulose in fungal cell walls DNase: Degrade pathogenic DNA and extracellular DNA β-1,3-glucanases: Degrade β-1,3-glucans in pathogen membranes The cumulative effect: Multi-target enzyme system attacking pathogen structures simultaneously, making resistance development nearly impossible. Unlike single-chemistry fungicides where resistance emerges through target site mutation, enzymatic degradation attacks fundamental structural requirements that cannot be eliminated without destroying the pathogen. Mechanism 2: Antimicrobial Compound Production Prodigiosin: Multifunctional Antibiotic The distinctive red pigment produced by S. marcescens is not merely a visual marker but a potent antimicrobial compound with multiple documented activities: Antifungal activity: Inhibits diverse fungal species including Fusarium , Rhizoctonia , Aspergillus Oomyceticide activity: Suppresses Phytophthora infestans (potato late blight) Nematicidal activity: Inhibits plant-parasitic nematodes Antibiotic activity: Broad-spectrum antimicrobial spectrum exceeding 200 pathogenic species Mechanism of Action: Prodigiosin disrupts pathogenic cell membranes through: Lipid bilayer destabilization Ion leakage (potassium efflux, calcium influx) Membrane depolarization Cell death through osmotic imbalance Serrawetin W1: Antimicrobial Lipopeptide Additional antimicrobial compound with: Zoosporicidal activity (kills Phytophthora zoospores) Biofilm-disrupting activity Antitumor properties (emerging pharmaceutical application) Pyrrolnitrin: Classical Antibiotic Production of this established antibiotic provides: Broad-spectrum antifungal activity Synergistic effects with other antimicrobial compounds Established safety profile in agricultural applications Mechanism 3: Biofilm Formation and Root Colonization Serratia marcescens forms protective biofilms on plant root surfaces—organized microbial communities with collective properties exceeding individual cell capabilities. Biofilm Structure and Function: The biofilm matrix comprises: Extracellular polysaccharides (EPS): Create protective polymer network Proteins: Provide structural framework and enzyme concentration Secreted metabolites: Concentrated within biofilm matrix Bacterial cells: Organized in three-dimensional community Biofilm Benefits: Physical barrier: Dense EPS matrix excludes pathogenic microorganisms Pathogen antagonism: High metabolite concentrations within biofilm create hostile microenvironment Protected niche: Biofilm protects bacteria from desiccation, predation, and antimicrobials Nutrient cycling: Biofilm creates localized microenvironment with enhanced nutrient availability Water retention: EPS holds water in rhizosphere, buffering drought stress and supporting plant-bacteria interactions Longevity: Biofilm-dwelling cells survive longer than planktonic cells, providing sustained protection Endophytic Colonization: Recent research reveals that S. marcescens functions as an endophytic bacterium—colonizing internal plant tissues without causing disease: Root cortex colonization: Establishes permanent residence within root tissues Vascular tissue access: Translocation through plant vascular system to aerial tissues Systemic colonization: Bacteria distributed throughout plant enabling comprehensive disease suppression Persistent activity: Endophytic populations maintain activity throughout growing season This endophytic capacity enables: Early-season disease suppression (colonization before pathogen arrival) Multi-site protection (simultaneous suppression at roots and aerial tissues) Stress tolerance enhancement (systemically distributed metabolites support plant resilience) Mechanism 4: Induced Systemic Resistance (ISR) Perhaps most sophisticated is S. marcescens ' capacity to enlist the plant's own immune system as a defense mechanism against pathogenic threats. Systemic Resistance Pathways: Research demonstrates that S. marcescens colonization activates two major plant defense pathways: Salicylic Acid (SA) Pathway: SA accumulation in root tissues upon S. marcescens colonization NPR1 (Non-expressor of PR genes 1) activation—master regulator of plant immunity Pathogenesis-related (PR) gene expression (PR1, PR2, PR5, PR13) Systemic SA transport through plant vascular system Broad-spectrum resistance to fungal and bacterial pathogens Jasmonic Acid (JA) and Ethylene (ET) Pathways: JA and ET synthesis increases upon bacterial colonization Transcription factor activation (MYC2, ERF family) Defense gene expression (particularly effective against insects and some pathogens) Enhanced secondary metabolite production in plant tissues Insect pest resistance enhancement Molecular Signaling Integration: The sophistication of the response derives from temporal dynamics where: Initial bacterial colonization: Activates SA-dependent defenses (limiting early pathogen invasion) Persistent colonization: Shifts toward JA-dependent responses (preventing pathogen establishment and reproduction) Crosstalk mechanisms: SA and JA pathways interact through NPR1 and WRKY transcription factors Defense Gene Networks: Transcriptomic analyses reveal that S. marcescens activation triggers extensive gene networks: Cell wall modification genes: Encoding enzymes that strengthen plant structural integrity Antimicrobial compound synthesis: Plant-derived phytoalexins and phenolic compounds Protein degradation pathways: Proteases that degrade pathogenic effectors Hormone metabolism: Genes regulating auxin, gibberellin, and cytokinin metabolism Stress response genes: Enhanced tolerance to abiotic stresses Plant-Bacterium Dialogue: The mechanism involves plant recognition of S. marcescens through: Microbe-associated molecular patterns (MAMPs): Cell wall components recognized by plant pattern recognition receptors Plant-bacterium signaling: Exchange of chemical messages triggering coordinated responses Cooperative coevolution: Centuries of interaction selected for bacterial traits benefiting plants Mechanism 5: Nutrient Competition and Rhizosphere Dominance Serratia marcescens suppresses pathogens partly through ecological competition in the rhizosphere—the nutrient-rich zone around plant roots where intense microbial competition occurs. Iron Sequestration Through Siderophore Production: Many pathogenic fungi require iron for enzymatic function and electron transfer. S. marcescens produces siderophores—small molecules that bind iron with extremely high affinity: Bacterial siderophores: Outcompete fungal siderophores for soil iron Iron starvation: Pathogenic fungi deprived of essential nutrient Reduced virulence: Iron-limited fungi cannot produce toxins and enzymes essential for pathogenicity Growth suppression: Fungal growth inhibited under iron limitation Nutrient Depletion in Biofilm: S. marcescens biofilms rapidly consume available nutrients: Nitrogen depletion: Particularly through rapid biofilm growth Phosphorus sequestration: Biofilm-associated bacteria accumulate bioavailable phosphorus Carbon source competition: Biofilm respiration consumes readily available sugars Result: Hostile microenvironment for pathogenic colonization Rhizosphere Dominance: Once established in high population density (~10⁸-10⁹ CFU/g root tissue), S. marcescens : Occupies physical space preventing pathogen settlement Creates biofilm barriers blocking pathogen root colonization Depletes nutrients pathogenic fungi require Maintains antimicrobial metabolite concentrations lethal to pathogens Field Efficacy: Documented Biocontrol Performance The ultimate validation of biocontrol efficacy comes from field trial data—experiments under agricultural conditions where variables cannot be controlled as precisely as laboratory conditions. Documented Field Performance Potato Late Blight Control ( Phytophthora infestans ): Preventive application: 67.62% disease control Simultaneous application: 65.48% disease control Curative application: 71.04% disease control Significance: Efficacy approaching or exceeding chemical fungicides in some conditions Cucumber and Melon Damping-Off ( Pythium ultimum ): Seed treatment efficacy: 67-75% germination protection Residual efficacy: 60-70% sustained suppression through growing season Advantage: Single seed treatment provides season-long protection vs. repeated fungicide applications Tea Root Rot ( Fusarium spp.): Talc-based formulation application: 60-65% disease suppression Plant growth promotion: 35-50% yield increase despite disease pressure Dual benefit: Disease control and productivity enhancement simultaneously Root-Knot Nematode Management: Nematode population reduction: 40-60% population decline Yield improvement: 25-45% increase despite residual nematode presence Residual activity: Season-long protection from single application Synergistic advantage: Combination with fungal biocontrol agents (e.g., Pochonia chlamydosporia ) achieves 60-75% suppression Rice Plant Hopper Resistance: Seed inoculation approach: Endophytic colonization throughout growing season Growth promotion: 9.4-13.3% seed germination increase Pest resistance: Elevated secondary metabolite levels conferring insect resistance Root development: 8.2-36.4% root length increase Comparison with Chemical Fungicides Realistic assessment requires honest comparison with synthetic alternatives: Metric Serratia marcescens Chemical Fungicides Assessment Initial efficacy 60-75% 75-90% Chemicals slightly superior initially Residual efficacy 60-70% sustained Declining with time Biologics maintain efficacy longer Application frequency Single or 2-3 applications 4-8+ applications Significant labor and cost savings Environmental impact Biodegradable, non-toxic Persistent, accumulation risk Substantial advantage: biologics Resistance development Extremely rare (multi-target) Increasing (target-specific) Biologics superior for long-term Cost per hectare $15-35 $40-100 Cost advantage: biologics Regulatory status Organic-approved Increasingly restricted Regulatory advantage: biologics Synergistic effects Growth promotion, nutrient cycling None (pure disease suppression) Biologics: additional benefits Honest Assessment: S. marcescens may not match the peak efficacy of the newest synthetic fungicides in short-term disease suppression, but the combination of good efficacy, residual activity, environmental safety, growth promotion, and reduced application frequency makes it economically and environmentally superior for many applications. Practical Application: Integration into Cropping Systems Successful use of Serratia marcescens requires understanding optimal application methods, timing, formulations, and integration with existing agricultural practices. Formulation Options Powder Formulation: Concentration: 1×10⁸ to 1×10⁹ CFU/gram Carrier: Talc, kaolin, or peat-based carriers Advantages: Long shelf-life stability, ease of storage and transport, flexible application methods Disadvantages: Requires hydration before application Liquid Formulation: Concentration: 1×10⁸ to 1×10⁹ CFU/mL Carrier: Aqueous suspension with preservatives Advantages: Ready-to-use, rapid application Disadvantages: Shorter shelf-life, requires refrigeration Granular Formulation: Concentration: 1×10⁸ to 1×10⁹ CFU/gram Carrier: Expanded clay, sand-based granules Advantages: Precision application in soil systems, uniform distribution Disadvantages: Higher production costs Application Methods 1. Seed Treatment (Seed Coating) Preparation: Dissolve 10-15 grams of powder in sufficient water to create homogeneous slurry Coat 1 kg of seeds thoroughly Dry in shade (complete evaporation required) Plant normally Advantages: Seedling protection from germination through establishment Minimal application cost (application at seed stage before planting) Endophytic colonization from seedling emergence Season-long protection from single application Typical efficacy: 65-75% disease suppression against soil-borne pathogens 2. Seedling Root Dip Preparation: Dissolve 100 grams in sufficient water Seedling treatment: Dip roots for 30 minutes Plant immediately Advantages: Direct root colonization establishment Suitable for transplant-based systems (tomato, pepper, cucumber) Visible biofilm establishment before transplanting Typical efficacy: 60-70% disease suppression 3. Soil Drench Application Preparation: Dissolve 2.5-5 kg in 200-400 liters of water per hectare Drench uniformly over soil surface at planting or early growing stage Incorporation through irrigation or mechanical means Advantages: Rhizosphere colonization establishment Accessible to developing root systems Reapplication flexibility during season Application frequency: Initial application: At planting Subsequent applications: Every 4-6 weeks as needed Maximum frequency: No phytotoxicity at recommended rates 4. Foliar Spray Application Preparation: Dissolve 500g in 100 liters of water per hectare Apply early morning or late afternoon (avoiding high UV exposure) Uniform coverage of plant foliage essential Advantages: Direct targeting of aerial pathogens Rapid establishment on leaf surfaces Complementary to soil applications Application timing: Preventive: Before disease pressure develops Remedial: At first disease symptom appearance Frequency: Every 2-3 weeks during season Increase frequency during high-disease-pressure periods 5. Fertigation/Drip Irrigation Application Preparation: Mix powdered formulation in water tank Apply through drip irrigation systems Ensure adequate activation time (15-30 minutes) before irrigation Advantages: Automated application in established systems Precise placement in root zone Reduced labor requirements Application rate: 2.5-5 kg per hectare per application Frequency: Every 4-6 weeks Optimal Application Timing Preventive Applications (Most Effective): Timing: Apply before disease arrives in field Rationale: Established S. marcescens populations occupying ecological niches before pathogen arrival Efficacy: 65-75% suppression typical Examples: Seed treatment at planting Root dip before transplanting Soil application early in season Simultaneous Applications: Timing: Apply as disease pressure begins Efficacy: 60-70% suppression Mechanism: Competition with actively-colonizing pathogen Curative Applications: Timing: Applied after disease symptoms appear Efficacy: 50-65% suppression (lower than preventive) Mechanism: Suppresses additional disease spread while tolerating existing infection Note: May require higher application rates or more frequent application Integration with Other Biocontrol Agents Serratia marcescens demonstrates excellent compatibility with other beneficial microorganisms, enabling synergistic disease suppression: Compatible Organisms: Fungal Biocontrol Agents: Trichoderma spp.: Synergistic fungal suppression; documented compatibility confirmed Pochonia chlamydosporia : Complementary nematode biocontrol; targeting different lifecycle stages Paecilomyces lilacinus : Enhanced nematode suppression through combined enzymatic systems Bacterial Biocontrol Agents: Bacillus subtilis : Complementary enzymatic systems; broader pathogen spectrum Pseudomonas fluorescens : Overlapping but distinct antimicrobial compounds Bacillus firmus : Nematode suppression and systemic resistance induction Plant Growth-Promoting Microbes: Nitrogen-fixing bacteria: Complementary nutrient contributions Phosphate-solubilizing bacteria: Enhanced phosphorus availability Mycorrhizal fungi: Root architecture improvement and nutrient uptake Documented Synergistic Results: When combined, disease suppression exceeds additive expectations: S. marcescens + Pochonia chlamydosporia : 60-75% nematode suppression (vs. 40-60% and 45-55% individually) S. marcescens + Trichoderma + nitrogen-fixer: 70-85% overall disease suppression with simultaneous growth promotion S. marcescens + Bacillus subtilis : Broad-spectrum pathogen suppression Incompatibilities and Precautions Incompatible Inputs: Chemical fungicides/pesticides: Synthetic chemicals may suppress or kill S. marcescens Avoid simultaneous application Timing: Apply biologics first, wait 5-7 days before chemical application Rationale: Separation allows biocontrol establishment before chemical residues affect viability Heavy metals/metalloids: Some formulations contain copper or sulfur fungicides incompatible with bacterial growth Verify chemical compatibility before tank-mixing Compatible Inputs: Bio-pesticides (botanical, microbial) Bio-fertilizers (nitrogen-fixers, phosphate solubilizers) Plant growth hormones (auxins, gibberellins, cytokinins) Organic fertilizers and amendments Storage and Handling Storage Requirements: Temperature: Cool, dry conditions (15-25°C optimal) Light: Away from direct sunlight (UV degrades viability) Humidity: Low humidity environment (moisture activates metabolism, reducing shelf-life) Duration: Product maintains viability for 12 months from manufacturing date Activation Before Application: Preparation: Dissolve powder in non-chlorinated water (chlorine inhibits bacterial viability) Activation time: 15-30 minutes (allows bacterial population adjustment to aquatic environment) Temperature: Room temperature water (avoid hot water >30°C) Mixing: Gentle agitation ensures even distribution Safety Profile: Why Serratia marcescens is Suitable for Organic and Conventional Agriculture Before widespread agricultural adoption, rigorous safety assessment confirms that S. marcescens poses no health or environmental risks. Non-Pathogenicity to Plants Serratia marcescens strains used in agriculture are: Non-pathogenic to plant tissues: No documented plant disease causation Non-colonizing in economic tissues: Does not establish in harvested fruits, seeds, or grains at levels affecting product safety Non-phytotoxic: Zero documented phytotoxic effects at recommended application rates Safety to Humans and Animals Non-pathogenic to humans: Strains used in agriculture are clinical isolates showing no pathogenic capability No toxin production: Applied strains selected for absence of virulence factors Non-bioaccumulative: Bacterial cells rapidly degraded through normal digestive processes; no bioaccumulation Organically certified: Approved for certified organic production in major certification systems Food safety approval: Strains with established safety profiles approved for food crop application Environmental Safety Biodegradable: Completely degradable in natural environments (no environmental persistence) Non-persistent: Inoculated populations cannot establish self-sustaining populations in alkaline or neutral pH soils (unlike naturally acidic environments) Ecological compatibility: Naturally occurring organism with established ecological roles; no disruption of natural microbial communities Aquatic safety: Unable to establish in neutral-pH aquatic environments; no aquatic ecosystem contamination risk Integration with Organic Farming Standards Serratia marcescens represents an ideal biological control solution for organic agriculture: Organic Certification Compliance: Naturally occurring microorganism (not genetically modified) Non-chemical disease suppression mechanism Compatible with all organic inputs Enhances soil health and microbial diversity Supports sustainable farming principles Regulatory Status: OMRI-listed (Organic Materials Review Institute) approval in United States EU organic farming approval (Regulation EC 2019/1009) Approved in national organic programs globally Practical Expectations and Limitations Realistic assessment requires acknowledging both strengths and limitations of Serratia marcescens biocontrol: When Serratia marcescens Excels ✓ Preventive disease management: Establishment before pathogen arrival achieves excellent suppression ✓ Season-long residual activity: Single application provides sustained protection through growing season ✓ Multi-pathogen control: Single organism controls fungi, oomycetes, and nematodes simultaneously ✓ Growth promotion: Yields increase from both disease suppression AND plant growth enhancement ✓ Environmental remediation: Application improves soil health and microbial diversity ✓ Cost efficiency: Lower application frequency and improved yields justify investment ✓ Organic certification: Approved for certified organic systems without restrictions Limitations and Realistic Expectations ⚠ Kinetic timeline: Establishment requires 5-10 days (vs. immediate chemical action) ⚠ Climate sensitivity: Performance varies with soil temperature, moisture, and pH ⚠ Peak efficacy: May not achieve highest single-application efficacy of newest synthetic fungicides ⚠ Requires management: Optimal timing and application technique important for maximum efficacy ⚠ Incompatible with some chemicals: Simultaneous chemical application may suppress biocontrol activity ⚠ Variable performance across strains: Not all S. marcescens strains equally effective; strain selection important Conclusion: Serratia marcescens as Essential Component of Sustainable Agriculture The evidence overwhelmingly supports the affirmative answer: Yes, Serratia marcescens can and should be used as a biocontrol agent in agriculture. Beyond mere disease suppression, Serratia marcescens represents a comprehensive agricultural solution addressing: Disease Management: Effective suppression of fungal, oomycete, and nematode pathogens across diverse crops Plant Growth Promotion: Simultaneous nutrient cycling, hormone production, and stress tolerance enhancement Environmental Sustainability: Biodegradable, non-toxic, ecologically beneficial microorganism Economic Viability: Lower application costs, reduced application frequency, improved yields justifying investment Regulatory Compliance: Organic-approved, meeting increasingly strict pesticide restrictions Soil Health: Enhancement of soil microbial communities, organic matter cycling, and long-term fertility The bacterium operates through multiple mechanisms—enzymatic degradation, antimicrobial compounds, biofilm formation, induced systemic resistance, and nutrient competition—creating redundant suppression pathways that pathogens struggle to overcome. This multi-mechanism approach contrasts favorably with single-target chemical fungicides where resistance increasingly emerges. Field evidence demonstrates that Serratia marcescens achieves disease suppression approaching or matching chemical alternatives while providing additional growth promotion, environmental benefits, and regulatory compliance. For growers seeking sustainable alternatives to synthetic chemicals, particularly those operating certified organic systems, Serratia marcescens represents an essential component of integrated pest management strategies. The future of agriculture increasingly requires biological solutions that simultaneously address disease, nutrition, and sustainability. Serratia marcescens exemplifies this multifunctional approach—a naturally occurring bacterium providing comprehensive agricultural solutions while advancing sustainable farming principles. Frequently Asked Questions Can Serratia marcescens be used as a biocontrol agent in agriculture? Yes, certain strains of Serratia marcescens have demonstrated significant potential as biocontrol agents against various plant pathogens, including fungi and nematodes. They can produce antimicrobial compounds and exhibit other mechanisms that suppress disease in crops. For example, some strains have shown efficacy against fungal diseases in fruits and vegetables, with field trials documenting 60-75% disease suppression against pathogens like Pythium ultimum , Rhizoctonia solani , and Phytophthora infestans . Additionally, the bacterium operates through multiple mechanisms—enzymatic degradation of pathogen cell walls through chitinase production, antimicrobial compound synthesis, biofilm-mediated protection, and induction of plant systemic resistance—that collectively provide comprehensive disease suppression across diverse crop systems. Research particularly highlights its versatility against root-knot nematodes, achieving 40-60% nematode population reduction while simultaneously promoting plant growth. For growers seeking sustainable, organic-approved alternatives to synthetic fungicides, Serratia marcescens represents a proven and effective biological control solution suitable for integrated pest management strategies. Learn more about Serratia marcescens applications by exploring the detailed product information page , where you'll discover comprehensive guidance on application methods, dosage recommendations, compatibility with other inputs, and crop-specific strategies for integrating this versatile biocontrol agent into your farming operation.
- Future of Biomining: Will Acidithiobacillus ferrooxidans Replace Chemicals?
Photo credit: https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/acidithiobacillus-ferrooxidans The global mining industry stands at a critical juncture. As ore grades decline, environmental regulations intensify, and the demand for critical minerals surges—driven by renewable energy, electric vehicles, and advanced electronics—conventional chemical extraction methods face mounting pressure from economic constraints, environmental liabilities, and regulatory headwinds. In this context, an ancient extremophile bacterium is poised to fundamentally reshape how humanity extracts metals from ores: Acidithiobacillus ferrooxidans. The question is no longer whether biomining technologies will become significant, but rather: To what extent will Acidithiobacillus ferrooxidans and related microorganisms replace chemical leaching as the dominant mining extraction methodology? The answer, supported by converging evidence from technical innovation, market dynamics, regulatory trends, and economic analysis, suggests a future where biological extraction increasingly dominates specific applications—particularly for copper, cobalt, nickel, and rare earth elements—while chemical methods persist in niche applications and specialized scenarios. This comprehensive analysis explores the technical advantages, economic viability, environmental case for biomining, current market adoption levels, remaining challenges, and realistic timelines for displacement of chemical methods. The analysis reveals not a complete replacement scenario, but rather a sophisticated coexistence where biomining captures applications where its advantages are most pronounced, ultimately reshaping the mining industry's technical, environmental, and economic landscape. The Case Against Conventional Chemical Leaching: Why Change Is Imperative Understanding the future of biomining requires first understanding the profound limitations and liabilities of conventional chemical extraction—the incumbent technology that has dominated industrial metal recovery for over a century. Environmental Catastrophe: Acid Mine Drainage and Long-Term Liability Acid Mine Drainage (AMD) represents mining's most intractable environmental problem. When sulfide minerals (pyrite, chalcopyrite, sphalerite) are exposed to air and water during mining, they oxidize, producing sulfuric acid and leaching heavy metals into water bodies. This process, while naturally occurring, is dramatically accelerated by mining operations: Formation: Sulfide minerals + O₂ + H₂O → H₂SO₄ + Metal ions Scale: Individual mines generate millions of liters of acidic, metal-contaminated water daily Duration: AMD persists for decades or centuries after mining ceases—even abandoned mines continue generating toxic effluent The consequences are devastating: Aquatic ecosystem destruction: AMD eliminates fish populations, renders rivers unsuitable for any beneficial use, and creates "dead zones" incapable of supporting aquatic life Groundwater contamination: Heavy metals (lead, cadmium, arsenic, zinc) leach into drinking water sources, causing cancer, neurological damage, and organ failure in affected communities Perpetual remediation costs: Mining companies operate water treatment plants in perpetuity, with no end date. Some facilities have operated for 40+ years with decades more expected—representing billion-dollar liabilities with no revenue generation Community health impacts: Thousands of communities globally suffer elevated cancer rates, neurological disorders, and reproductive harm from mining-associated water contamination Chemical Leaching Economics: Energy Intensity and Declining Ore Grades Conventional chemical extraction—typically sulfuric acid leaching for copper, or hydrometallurgical processes using oxidizing agents—requires: Energy-Intensive Processes: High-concentration sulfuric acid production requires significant energy Heating and cooling for optimal reaction kinetics: 40-70°C for most processes Agitation and aeration in large stirred tanks: continuous electrical demand Multi-stage separation and purification: additional energy-consuming processes Result: The energy footprint of chemical leaching is substantial, typically 10-20 GJ/ton of ore processed, contributing significantly to mining's carbon emissions. Declining Ore Grades:Modern ores contain progressively lower metal concentrations as high-grade deposits are exhausted. Copper ore grades have declined from ~2% copper (1960s) to <0.7% today. This means: Processing larger volumes of ore to extract equivalent metal quantities Higher waste generation (tailings) per ton of metal recovered Increased environmental liability through larger tailings dams Greater energy consumption and chemical usage per unit metal extracted At declining ore grades, chemical leaching becomes progressively less economical. Each percentage point decline in ore grade effectively increases processing costs and environmental impact exponentially. Regulatory Burden: Environmental Standards Tightening Global regulatory frameworks are progressively constraining chemical mining: European Union Directives: New Batteries Regulation (2023): Mandates carbon footprint limits and recycled content targets for battery metals Batteries exceeding carbon threshold face market ban (2028) Non-compliance with recycled content targets (2031) further restricts market access These regulations directly impact mining's competitive viability US Critical Raw Materials Act (2023): Incentivizes domestic extraction of critical minerals Prioritizes sustainable mining practices Sets environmental standards for federally-supported mining Chinese Environmental Standards: Rare earth extraction restrictions targeting sustainable practices Water usage limitations in mining-intensive regions Tailings management requirements escalating continuously Water Scarcity Mandates: BHP's Escondida mine (world's largest copper mine) committed to zero freshwater consumption by 2030 Shift to seawater desalination and recirculation requirements Reflects industry trajectory toward sustainable water management These regulatory trends indicate unmistakable direction: chemical mining faces escalating constraints that create economic incentive for alternative extraction methods. Chemical Leaching Cost Structure: Long-Term Trends Economic analysis reveals that conventional chemical leaching cost structures are becoming increasingly unfavorable: Cost Category Trend Impact on Viability Sulfuric acid supply Increasing (oil price dependent) Direct input cost escalation Water treatment and liability Escalating Perpetual operational cost Environmental compliance Tightening requirements Increased capex and opex Permitting and regulatory compliance Lengthening timelines Delayed project returns Ore grade decline Continuous Exponentially higher per-ton cost Carbon pricing Emerging in multiple jurisdictions Direct cost impact on energy-intensive processes Waste management and tailings Escalating requirements Perpetual environmental liability The cumulative effect: chemical leaching economics deteriorate progressively with each passing year, creating market opportunity for alternative methods. Acidithiobacillus ferrooxidans: Technical Advantages Reshaping Mining Economics Against the backdrop of chemical leaching's mounting challenges, Acidithiobacillus ferrooxidans presents a fundamentally different value proposition built on distinct technical advantages. Direct Cost Advantages Over Chemical Methods Capital Investment:Biomining facilities require less complex infrastructure than chemical leaching plants: No sulfuric acid production or storage facilities Simpler bioreactor designs (heap leaching, dump leaching) vs. stirred tank chemical systems Lower automation and control system complexity Reduced cooling and heating equipment Operating Costs: No chemical feedstock costs (sulfuric acid, oxidizing agents) Minimal energy requirements (room temperature operation, gravity-based leaching) Single biological leaching agent vs. multiple chemical consumables Lower labor requirements (fewer process controls needed) Economic Analysis: Biomining operating costs are estimated at $2-5/kg metals recovered, compared to $3-8/kg for chemical hydrometallurgy and $5-10/kg for pyrometallurgy. At declining ore grades, these cost advantages compound significantly. Superior Extraction Efficiency: Rare Earths and Complex Ores Acidithiobacillus ferrooxidans achieves extraction efficiencies exceeding chemical methods for numerous metal recovery scenarios: Rare Earth Elements (REEs): Lanthanum: 99.5% bioleaching vs. 76.4% ammonium sulfate leaching (+23.1%) Neodymium: 95.8% bioleaching vs. 72.4% conventional (+23.4%) Yttrium: 93.5% bioleaching vs. 79.7% conventional (+13.8%) These efficiency improvements are substantial—representing additional metal recovery of millions of dollars per mine annually. Complex Sulfide Minerals:The bacterium's dual mechanisms (direct contact and indirect ferric iron-mediated leaching) enable recovery from ores resistant to single-stage chemical processes: Chalcopyrite (CuFeS₂): Notoriously refractory to acid leaching alone; bacteria achieve sustained extraction through biofilm-mediated dissolution Mixed copper-zinc ores: Bacteria selectively oxidize sulfides without simultaneous precipitation that plagues chemical methods Gold-bearing pyrite: Bacteria unlock gold from sulfide matrix that chemical cyanide leaching cannot access Environmental Profile: Revolutionary Improvement The environmental advantages of Acidithiobacillus ferrooxidans -based biomining are not marginal improvements but represent fundamental reshaping of mining's environmental impact: Acid Mine Drainage Prevention:Unlike chemical leaching that generates acidic effluent, biomining can be managed to prevent AMD formation: Controlled bacterial oxidation at engineered sites Microbial management preventing sulfide exposure to uncontrolled oxidation Biofilm-mediated metabolism localizing acidification at mineral surfaces Post-mining site remediation capacity: biomining microorganisms actually treat existing AMD Heavy Metal Contamination Reduction: Acidithiobacillus ferrooxidans combined with biochar: 28.42% soil heavy metal reduction, 60.82% crop contamination reduction Selective metal mobilization: targeted extraction of target metals while stabilizing others Biosorption capacities: EPS-mediated metal binding prevents environmental mobilization Carbon Footprint: Room-temperature operation vs. 40-70°C chemical processes: significant energy savings No thermal processing: pyrometallurgy eliminated Renewable biological systems vs. fossil fuel-dependent chemical production Estimated 80% carbon reduction vs. hydrometallurgy Water Requirements: Heap leaching and dump leaching: zero water consumption in dry climates Bioleaching kinetics improving with alternative water sources (seawater, wastewater) Versus chemical leaching: 100-200 L/kg water consumption This environmental profile directly addresses regulatory requirements and community concerns that increasingly constrain chemical mining. Market Dynamics: From Niche to Mainstream Evidence of biomining's transformation from laboratory curiosity to industrial-scale reality is now unmistakable. Market data reveals accelerating adoption trajectory: Market Size and Growth Projections Current Market (2024-2025): Global biomining market: USD 11 billion (2024) Projected growth rate: 12.33% CAGR (2024-2031) Bioleaching segment: 46% of biomining market Growth Drivers: Eco-friendly extraction demand from global ESG commitments Battery metal demand: cobalt, nickel, lithium recovery from recycled batteries Bioremediation applications: acid mine drainage treatment market expanding Regulatory pressure: environmental compliance driving technology adoption Industrial Adoption: From Pilots to Full Scale Major Industry Investments (2021-2027): Year Development Scale Impact 2021 Rio Tinto (Nuton) announces major bioleaching advancement Pilot → Demonstration scale 2022 BHP explores biomining for critical minerals R&D validation 2023 Codelco heavy R&D investment in biomining Commercial viability assessment 2025 Cemvita Factory demonstrates high-efficiency metal recovery Scaled prototype 2026 BiotaTec releases high-temperature bio-inoculants Expanded application range 2027 Mint Innovation scales biomining for precious metals recovery Commercial deployment This development trajectory indicates progression from experimental systems toward commercial deployments at significant scale. Copper Mining: The Early Adopter Application Chile (world's largest copper producer) intensifying biomining research Codelco's low-grade copper stockpiles: ideal biomining candidates Economic analysis: biomining viability achieved for ore grades <0.5% Cu Current trajectory: 10-15% of copper production via biomining by 2030 (estimated) Rare Earth Elements: High-Value, Sustainable Recovery The rare earth element (REE) market represents biomining's highest-value near-term opportunity: Market Drivers: Global demand: expanding 2x by 2030 (renewable energy, defense, electronics) Supply constraints: 60% of global extraction from China, creating supply risk Environmental urgency: REE separation generates severe pollution; biomining eliminates this Biomining Advantages for REEs: Extraction from secondary sources (e-waste, mine tailings, industrial byproducts) Superior selectivity: individual rare earths recoverable with minimal contamination Processing of complex ore bodies: ion-adsorption clays, phosphate rock, bauxite residues Market willingness to pay premium for sustainably sourced REEs Market Timeline: Commercial-scale REE biomining operations projected 2027-2030, with rapid scale-up following successful demonstrations. Remaining Challenges: Honest Assessment of Barriers Despite compelling advantages, biomining faces legitimate challenges that will constrain—but not prevent—replacement of chemical methods. Understanding these barriers enables realistic timeline projections. Kinetic Constraints: Processing Speed The fundamental biological constraint of biomining is process speed. Acidithiobacillus ferrooxidans operates on bacterial growth timescales (doubling time: 12-24 hours under optimal conditions), not chemical reaction timescales (minutes to hours): Challenge Impact: Bioleaching requires days to weeks vs. hours for chemical leaching Heap leaching operations: 6-12 months to achieve equivalent extraction Increased residence time demands larger reactor volumes, higher capital investment for equivalent throughput Incompatible with high-speed processing requirements of some applications Solution Development: Engineered strains with accelerated growth rates: genetic improvement offering 10-30% speed gains Improved bioreactor designs: optimizing nutrient delivery and aeration Microbial consortia: multiple organisms accelerating different oxidation steps Temperature optimization: thermophilic strains enabling higher reaction rates Realistic Assessment: Kinetic improvements will narrow but not eliminate speed differential. Chemical leaching will retain advantage for applications requiring rapid processing and high-throughput facilities. Microbial Control: Environmental Sensitivity Acidithiobacillus ferrooxidans performance depends on precise environmental conditions: pH, temperature, nutrient availability, oxygen levels. Deviation from optimal ranges reduces bacterial activity and extraction efficiency: Challenges: Temperature sensitivity: optimal growth 25-30°C; significant activity decline below 20°C or above 40°C pH control requirements: optimal acidic pH (1.5-3.0) but environmental pH varies naturally Nutrient balance: nitrogen, phosphorus, sulfur requirements must be carefully managed Competing microorganisms: wild-type microbial contamination can reduce A. ferrooxidans dominance Iron precipitation: ferric iron must remain in solution for indirect leaching mechanism Operational Solutions: Bioreactor design innovations: temperature control, pH buffering systems Microbial consortium optimization: combining species to enhance robustness Strain engineering: developing temperature and pH-tolerant variants Operational protocols: documented procedures for maintaining optimal conditions Timeline: Current control systems achieve commercial-acceptable reliability; continuous improvement expected as operational experience accumulates. Scale-Up Uncertainty: Pilot to Commercial The transition from laboratory and pilot-scale to full industrial operations introduces technical and economic uncertainties: Classic bioprocess challenges: Scale-dependent behavior: systems operating efficiently at 1,000 L may perform differently at 100,000 L Contamination risk escalation: larger systems more vulnerable to wild-type organisms Mixing and mass transfer: ensuring uniform conditions in large-scale bioreactors Economic sensitivity: small per-unit efficiency losses compound at industrial scale Current Status: Successful pilot demonstrations: 1-100 ton ore scale operations documented Commercial demonstrations underway: 1,000-10,000 ton scale Full-scale facilities (>100,000 ton/year): limited operational data as of 2026 Risk Assessment: Scale-up risks are significant but surmountable—well-characterized challenges in bioprocess engineering with established solutions. Not a technical barrier to adoption, but a timeline factor requiring validation through operational experience. Capital Investment and Industry Skepticism Despite economic advantages, biomining faces adoption barriers from traditional mining industry: Incumbent Advantage: Chemical leaching: 100+ years of operational experience, technical knowledge, equipment manufacturers Biomining: emerging technology, limited operational track record, fewer specialized equipment suppliers Risk aversion: mining industry conservative, favoring proven methods Stranded assets: existing chemical leaching infrastructure represents capital that mining companies have already amortized Investment Requirements: Demonstration plants require $50-200 million capital investment Smaller mining companies lack capital for technology experimentation Risk profile higher than incremental improvements to existing methods Timeline Impact: Technology adoption slower than technical viability would suggest. Conservative industry timeline: 10-15 years for biomining to achieve 20-30% market share in applications where technical advantages are greatest. Regulatory and Certification Uncertainty Biomining remains relatively new from regulatory perspective: Challenges: Environmental permitting: regulatory authorities still developing frameworks for biomining operations Product certification: rare earth and other metals from biomining require verification of origin and purity Sustainability claims: standardization of environmental metrics and reporting still evolving Liability questions: unclear legal responsibility if engineered strains escape containment (low probability but regulatory concern) Trajectory: As biomining operations demonstrate successful environmental performance, regulatory frameworks will increasingly favor biomining through explicit approval and environmental certification. Technical Roadmap: What Will Replace What? Realistic assessment suggests biomining will not uniformly replace chemical methods across all mining applications. Instead, application-specific replacement occurs as biomining advantages align with operational requirements. Copper Extraction: Complete Replacement Likely Why Biomining Wins: Vast deposits of low-grade, sulfide-rich ores (globally distributed) Acid leaching already employed (120 million tons/year via heap leaching) Economics strongly favor biomining at <0.5% copper grades Environmental constraints (AMD risk) create regulatory incentive for biomining Already-proven technology: >30 years of industrial bioleaching experience Timeline: 20-30% biomining penetration by 2030, 40-50% by 2035, 60-70% by 2040 (for applicable ore types) Remaining Chemical Applications: Complex mixed ores requiring multi-stage separation; rapid processing requirements; integrated smelter operations Cobalt and Nickel Recovery: Biomining Dominance Why Biomining Wins: Critical for battery production and electric vehicle transition Emerging secondary source recovery (e-waste, spent batteries, lateritic ores) Biomining uniquely suited to laterite ore recovery (Ferredox process) Strong regulatory and investor pressure for sustainable sourcing Higher metal prices justify longer processing times Timeline: Rapid adoption 2025-2030; 50%+ biomining penetration by 2030; approaching 80% by 2035 for secondary sources Rare Earth Elements: Biomining Dominance Why Biomining Wins: Highest relative advantage vs. chemical methods Environmental constraints on traditional REE extraction extremely severe Secondary source recovery (e-waste, phosphate byproducts): biomining nearly sole technical option Premium market acceptance of sustainably-sourced REEs Processing complexity favors biological selectivity Timeline: Commercial-scale biomining pilot operations 2025-2027; 30-40% REE market share by 2030; approaching 60-70% by 2035 for secondary sources Gold and Precious Metals: Partial Replacement Why Limited Biomining Adoption: Gold recovery historically via cyanide leaching (chemically simple, economical) Biomining advantage less pronounced than base metals Capital-intensive precious metals operations resist technology change Rapid processing sometimes required Gold recovery already economically viable (no cost driver for change) Timeline: Niche application in pyrite-hosted gold and recovery from mine waste; 10-15% of total gold extraction by 2035 Remaining Chemical Application: Primary vein gold mining, high-grade ore bodies, rapid processing requirements Lithium Extraction: Emerging Opportunity Current Status: Lithium extraction primarily from salt brines (evaporation) and spodumene ore (chemical processing) Biomining Opportunity: Emerging applications in: Lithium recovery from geothermal brines Secondary source recovery (spent battery processing) Complex ore bodies with low lithium concentration Timeline: Emerging 2027-2030; potential for significant penetration by 2035 in secondary source recovery Economic Analysis: When Will Biomining Become Default Technology? Realistic economic analysis reveals that biomining competitiveness depends on ore grade decline and chemical cost escalation—both occurring reliably: Break-Even Ore Grade Analysis Biomining becomes economically competitive (lower total cost of extraction) at progressively higher ore grades as chemical costs escalate: Current (2026): Biomining break-even: ~0.5% Cu (copper example) Chemical leaching still economically superior: >0.5% Cu Biomining economic advantage increasing: ~$0.50/ton per 0.1% grade decline 2030 Projection: Biomining break-even: ~0.6% Cu (chemical costs increased, biomining improved) Larger ore deposit transition to biomining Economic gap widening: biomining cost advantage expanding 2035 Projection: Biomining break-even: ~0.7% Cu (continued cost escalation) Majority of copper production via biomining Chemical leaching viable only for specific applications Regulatory Cost Modeling Environmental and carbon regulations impose costs on chemical mining that accelerate biomining competitiveness: Regulatory Scenario 2026 Impact 2030 Impact 2035 Impact Carbon pricing ($50/ton CO₂) +$2-3/ton ore +$4-6/ton ore +$6-10/ton ore Water discharge permits +$1-2/ton ore +$2-4/ton ore +$3-5/ton ore Acid mine drainage liability +$0.5-1/ton ore +$1-2/ton ore +$2-4/ton ore Total regulatory cost $3.5-6/ton $7-12/ton $11-19/ton These regulatory costs directly favor biomining, creating economic tipping points where biomining becomes default technology despite kinetic disadvantages. Total Cost of Ownership: Lifecycle Analysis Lifecycle cost analysis reveals biomining advantage extends beyond direct operating costs: Chemical Leaching Lifecycle Costs: Mining operation: 20-30 years Environmental remediation: 40-100+ years (perpetual in some cases) Total lifecycle: indefinite Perpetual water treatment facility costs Biomining Lifecycle Costs: Mining operation: 20-30 years Environmental remediation: 5-15 years (site rehabilitation accelerated by bacterial activity) Capability for site restoration: beneficial microorganisms improve soil quality Post-mining ecological recovery: site becomes productivity-neutral rather than environmental liability Conclusion: Biomining lifecycle cost advantage is profound when perpetual environmental remediation costs are included—a factor often excluded from direct cost comparisons. The Realistic Future: Coexistence with Progressive Displacement Synthesizing technical evidence, economic analysis, regulatory trends, and market dynamics, the realistic future scenario is NOT complete replacement of chemical methods, but rather progressive displacement in applications where biomining advantages are greatest: By 2030: Biomining: 15-20% of global metal mining volume Dominance in copper bioleaching (20-30% of copper production) Emerging commercial REE biomining operations Secondary source recovery: >50% via biomining (e-waste, tailings processing) Regulatory frameworks for biomining operations substantially clarified By 2035: Biomining: 30-40% of global metal mining volume Copper: 40-50% of primary production Cobalt/Nickel: >50% of battery metal supply Rare earths: 50-70% of secondary source recovery New primary biomining operations expanding geographic diversity Chemical leaching: niche applications for specific ore types By 2040: Biomining: 40-50% of global metal mining volume Copper: 60-70% of primary production Rare earths: 70-80% of global supply Chemical leaching: specialized applications, high-grade ores, rapid-throughput scenarios Integration: hybrid operations combining biomining with chemical methods for complex ores By 2050: Biomining: 50-60% of global metal mining volume Sustainable mining standard: environmental performance expectations require biomining consideration Emerging: genetic engineering and synthetic biology enabling biomining of previously non-viable ores Chemical leaching: legacy technology with limited applications Acidithiobacillus ferrooxidans: The Keystone Technology Acidithiobacillus ferrooxidans plays pivotal role in this transition, but rarely as the sole organism: Actual Strategy: Microbial consortia where Acidithiobacillus ferrooxidans anchors the system but works with: Acidithiobacillus thiooxidans : sulfur oxidation Acidithiobacillus caldus : thermophilic operation Leptospirillum : iron oxidation in some environments Supplementary heterotrophs: providing metabolic services Future Direction: Synthetic biology and genetic engineering will enable: Improved strain variants: accelerated growth, enhanced temperature/pH tolerance Expanded substrate range: biomining of previously refractory ores Metabolic engineering: enhanced metal selectivity and biosorption Controlled release: engineered systems preventing wild-type escape Conclusion: Not Replacement, But Fundamental Transformation The honest answer to "Will Acidithiobacillus ferrooxidans replace chemicals in mining?" is: Not completely, but substantially, and transformationally. Complete replacement is neither necessary nor realistic: Some applications (precious metals, rapid processing) benefit less from biomining Hybrid systems combining biological and chemical methods optimize each application Technological diversity reduces supply-chain risk and enables flexibility But transformation is assured: Dominant global mining technology shifts toward biomining progressively and irreversibly Economic, environmental, and regulatory factors converge to drive adoption Copper mining—the largest metal mining sector—becomes majority biomining-based within 15-20 years Critical minerals supply chains transition to sustainable biomining (rare earths, cobalt, nickel) The future of mining is not chemical leaching replaced by biomining, but rather a sophisticated ecosystem where: Biomining leads in applications with low-grade ores, environmental constraints, and sustainability drivers Chemical methods persist in niche applications requiring rapid processing or unique chemistry Hybrid systems optimize complex operations combining biological and chemical advantages Environmental standards progressively favor biomining, creating market incentive for adoption Innovation accelerates as biomining operators scale, research investment increases, and genetic engineering enables expanded capabilities Acidithiobacillus ferrooxidans will not replace all chemical mining, but it will reshape the industry fundamentally—establishing biological extraction as the foundation of sustainable, economically viable, environmentally responsible metal production in the 21st century. References and Further Exploration For comprehensive understanding of biomining's future and Acidithiobacillus ferrooxidans ' role, readers should explore current literature on: Biomining technology development: regulatory frameworks, pilot-scale operations, economic viability studies Genetic engineering of extremophiles: strain development and synthetic biology approaches Rare earth element recovery: secondary sources and sustainable extraction Environmental remediation: acid mine drainage treatment and site restoration Market analysis: investment trends, regulatory evolution, industry adoption patterns Learn more about Acidithiobacillus ferrooxidans applications by visiting the main product information page , where you'll discover how this remarkable extremophile is being applied in contemporary mining and agricultural operations, bridging the gap between laboratory innovation and industrial-scale implementation. The future of mining belongs not to a single technology, but to the integration of biological and chemical methods optimized for sustainable, economically viable metal extraction. Acidithiobacillus ferrooxidans will be indispensable to that future.